Chapter 1: 17P (page 75)
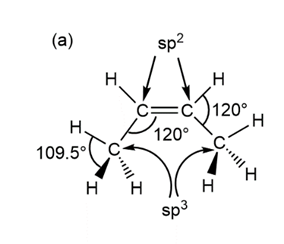
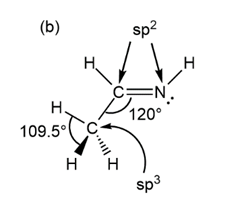
Predict the hybridization, geometry, and bond angles for the central atoms in (a) but-2-ene, CH3CH=CHCH3 (b)CH3CH=NH .
Short Answer
Learning Materials
Features
Discover
Chapter 1: 17P (page 75)
Predict the hybridization, geometry, and bond angles for the central atoms in (a) but-2-ene, CH3CH=CHCH3 (b)CH3CH=NH .


All the tools & learning materials you need for study success - in one app.
Get started for free
Give the molecular formula of each compound shown in Problem 1-37.

Question: Repeat Problem, this time drawing line-angle structures for compounds (a) through (h)
Question: Draw one valid Lewis structure for each compound. Assume the atoms are arranged as drawn
a,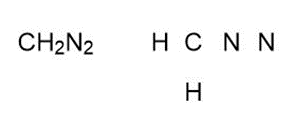
b.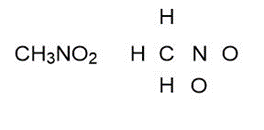
c.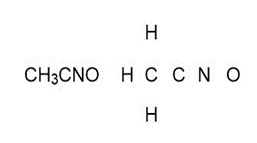
d.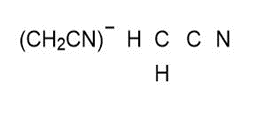
In pent− 2 −yne (CH3CCCH2CH3), there are four atoms in a straight line. Use dashed lines and wedges to draw three-dimensional representation of this molecule, and circle the four atoms that are in a straight line.
What do you think about this solution?
We value your feedback to improve our textbook solutions.