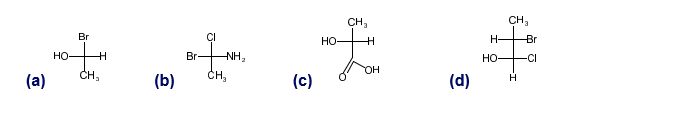
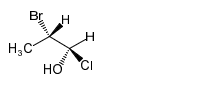
Chapter 5: Q-5-29P (page 280)
Convert the following Fischer projections to perspective formulas.
Short Answer
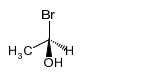
(a)
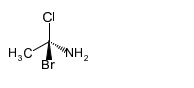
(b)
(c)
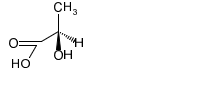
(d)
Learning Materials
Features
Discover
Chapter 5: Q-5-29P (page 280)
Convert the following Fischer projections to perspective formulas.

(a)

(b)

(c)
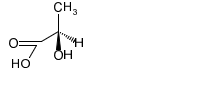
(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
For each Fischer projection, label each asymmetric carbon atom as (R) or (S). (a)-(f) the structures in Problem 5-18
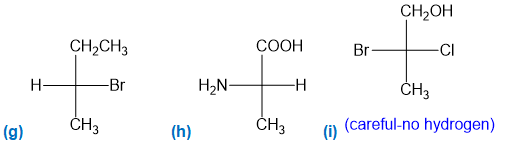
Question. For each structure,
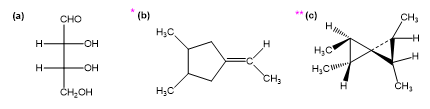
Question: Draw all distinct stereoisomers for each structure. Show the relationships (enantiomers, diastereomers, etc.) between the isomers. Label any meso iosmers, and draw any mirror planes of symmetry.
(a) CH3 -CHCI-CHOH-COOH
(b) tartaric acid, HOOC-CHOH - CHOH-COOH
(c) HOOC -CHBr-CHOH- CHOH-COOH
(d)
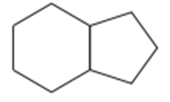
(e)
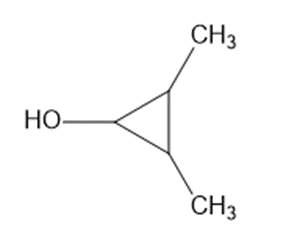
The following structures are optically active compounds. Star (*) the asymmetric carbon atoms in these structures.
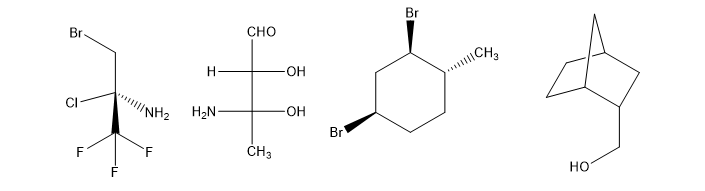
To show that (R)-2-butyl (R,R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.
What do you think about this solution?
We value your feedback to improve our textbook solutions.