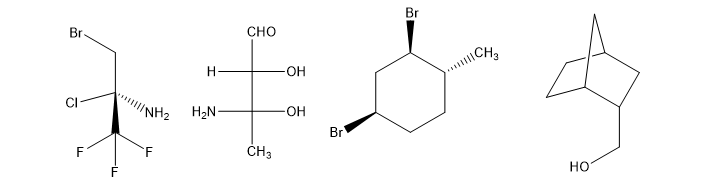
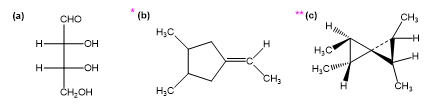
Chapter 5: Q19P (page 266)
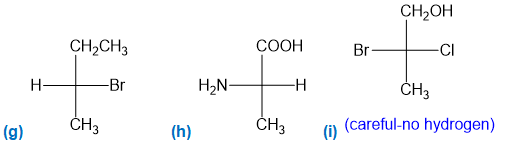
For each Fischer projection, label each asymmetric carbon atom as (R) or (S). (a)-(f) the structures in Problem 5-18
Short Answer
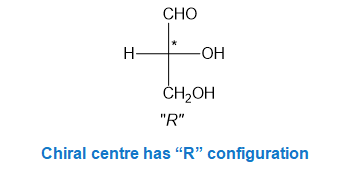
The asymmetric carbon atoms have been labelled and assigned the absolute configurations, (R) and (S).
Step by step solution
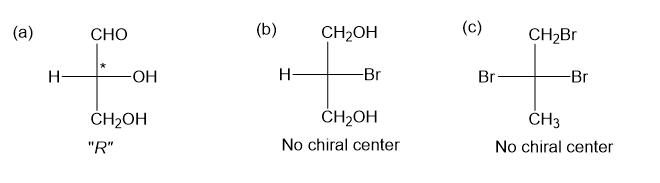
Step-1. Explanation of part (a):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page. By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centers are designated as “��”or ���”.
In part (a), the molecule is chiral and have “��” configuration.
Step-2. Explanation of part (b):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (b), the molecule is not chiral as it has no chiral centre present, thus, no configuration can be assigned.
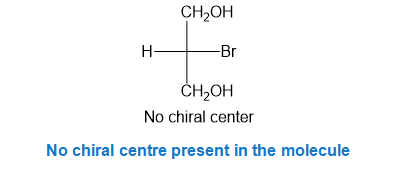
Step-3. Explanation of part (c):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (c), the molecule is not chiral as it has no chiral center present, thus no configuration can be assigned.
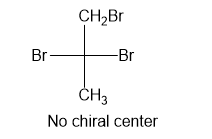
No chiral centre present in the molecule
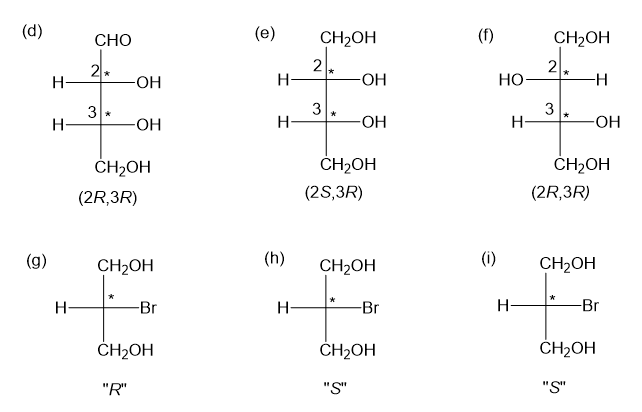
Step-4. Explanation of part (d):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (d), the molecule is chiral and has two chiral centres and configurations have been assigned to the two chiral centres.
Step-5. Explanation of part (e):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (e), the molecule is chiral with two chiral centres and configurations have been assigned to the two chiral centres.
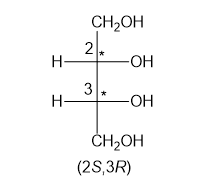
Chiral centres have (2S,3R) configuration
Step-6. Explanation of part (f):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
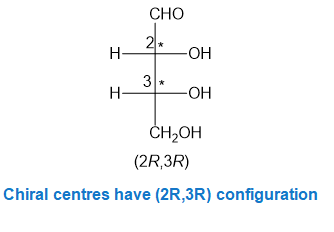
In part (f), the molecule is chiral with two chiral centres and configurations have been assigned to the two chiral centres.
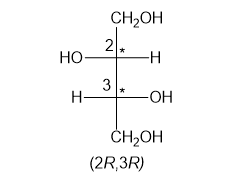
Chiral centres have (2R,3R) configuration
Step-7. Explanation of part (g):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (g), the molecule is chiral and chiral centre has “��” configuration.
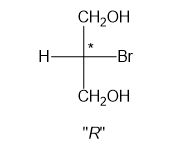
Chiral centre has “��” configuration
Step-8. Explanation of part (h):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (h), the molecule is chiral and chiral centre has “S” configuration.
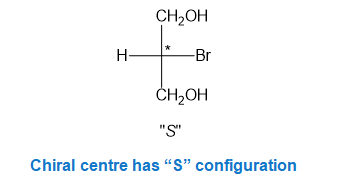
Step-9. Explanation of part (i):
Fischer projection is a method of representing the three-dimensional structures of molecules in two-dimensional form on a page.By convention, horizontal lines represent bonds projecting from the plane of paper towards the viewer and vertical lines represent bonds projecting away from the viewer. Absolute configuration is the arrangement of atoms or group of atoms that are independent of any other atom or group of atoms in the molecule. Chiral centres are designated as “��”or ���”.
In part (i), the molecule is chiral and chiral centre has “S” configuration.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!