Chapter 5: Question 5.33 (page 281)
(+)-Tartaric acid has a specific rotation of +12.0°. Calculate the specific rotation of a mixture of 68% (+)@tartaric acid and 32% (-)@tartaric acid.
Short Answer
Answer:
Specific rotation = 4.3
Learning Materials
Features
Discover
Chapter 5: Question 5.33 (page 281)
(+)-Tartaric acid has a specific rotation of +12.0°. Calculate the specific rotation of a mixture of 68% (+)@tartaric acid and 32% (-)@tartaric acid.
Answer:
Specific rotation = 4.3
All the tools & learning materials you need for study success - in one app.
Get started for free
2.Star (*) each asymmetric carbon atom in part (1), label each as (R) or (S) and compare your result from part (1) with the prediction you would make based on the asymmetric carbons.
To show that (R)-2-butyl (R,R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.
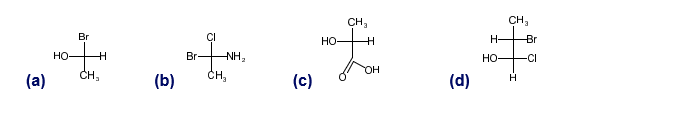
Convert the following Fischer projections to perspective formulas.
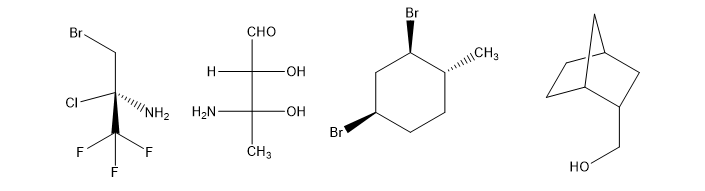
The following structures are optically active compounds. Star (*) the asymmetric carbon atoms in these structures.
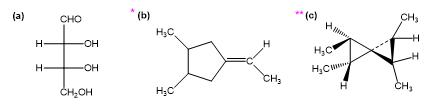
Question. For each structure,
What do you think about this solution?
We value your feedback to improve our textbook solutions.