Chapter 25: Q-25-4P (page 1308)
Give equations to show the reactions of sodium myristate with the following.(a) Ca2+ (b) Mg2+ (c) Fe3+
Short Answer
(a)
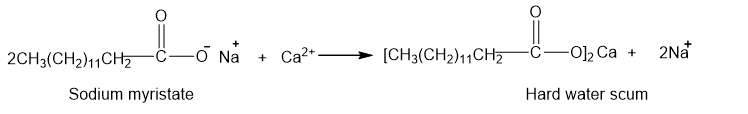
(b)
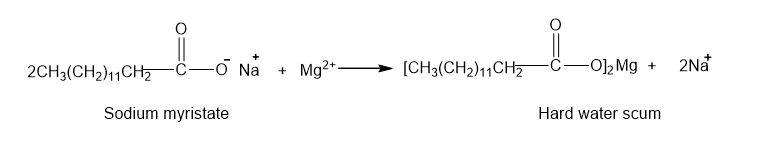
(c)
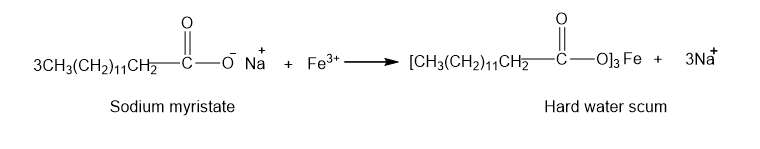
Learning Materials
Features
Discover
Chapter 25: Q-25-4P (page 1308)
Give equations to show the reactions of sodium myristate with the following.(a) Ca2+ (b) Mg2+ (c) Fe3+
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Circle the eight isoprene units in β-carotene.
Point out the hydrophilic and hydrophobic regions in the structures of sodium dodecyl sulfate, Nonoxynol®, and the alkylbenzenesulfonate detergent (Figure 25-6).
Question: Trimyristin, a solid fat present in nutmeg, is hydrolyzed to give one equivalent of glyceroland three equivalents of myristic acid. Give the structure of trimyristin.
Draw the structure of an optically active triglyceride containing one equivalent of stearic acid and two equivalents of oleic acid. Draw the products expected when this triglyceride reacts with the following reagents. In each case, predict whether the products will be optically active.
The synthesis of the alkylbenzenesulfonate detergent shown in figure 25-6 begins with the partial polymerization of propylene to give a pentamer.
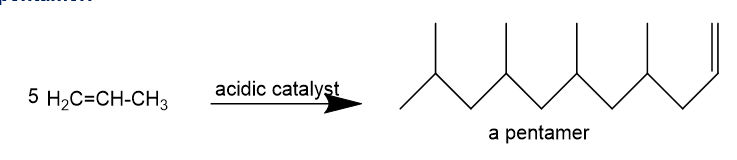
Show how aromatic substitution reactions can convert this pentamer to the final synthetic detergent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.