Chapter 25: Q1P. (page 1303)
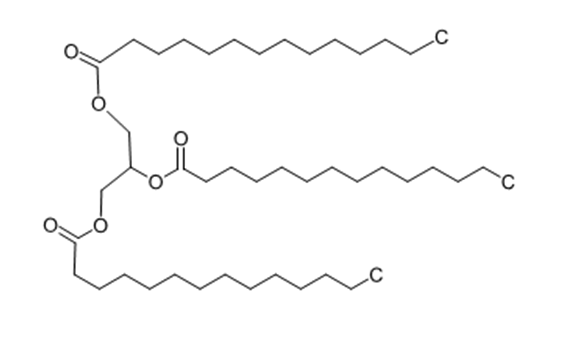
Question: Trimyristin, a solid fat present in nutmeg, is hydrolyzed to give one equivalent of glyceroland three equivalents of myristic acid. Give the structure of trimyristin.
Short Answer
Learning Materials
Features
Discover
Chapter 25: Q1P. (page 1303)
Question: Trimyristin, a solid fat present in nutmeg, is hydrolyzed to give one equivalent of glyceroland three equivalents of myristic acid. Give the structure of trimyristin.

All the tools & learning materials you need for study success - in one app.
Get started for free
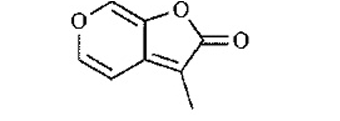
Two naturally occurring lactones are shown. For each compound, determine
(a) Whether the compound is a terpene. If so, circle the isoprene units.
(b) Whether the compound is aromatic and explain your reasoning.
(c) Show the product resulting from saponification with aqueous.
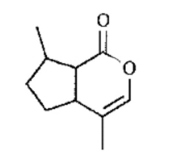
nepetalactone - the main ingredient in catnip
This compound generated in the smoke from burning plants, promotes seed germination in plants that require fire to reproduce.
When an extract of parsley seed is saponified and acidified, one of the fatty acids isolated is petroselenic acid, formula C18H34O2. Hydrogenation of petroselenic acid gives pure stearic acid. When petroselenic acid is treated with warm potassium permanganate followed by acidification, the only organic products are dodecanoic acid and adipic acid. The NMR spectrum shows absorptions of vinyl protons split by coupling constants of 7 Hz and 10 Hz. Propose a structure for petroselenic acid, and show how your structure is consistent with these observations.
Phospholipids undergo saponification much like triglycerides. Draw the structure of a phospholipid meeting the following criteria. Then draw the products that would result from its saponification.
(a) a cephalin containing stearic acid and oleic acid
(b) a lecithin containing palmitic acid.
Which of the following chemical reactions could be used to distinguish between a polyunsaturated animal oil and a synthetic cooking oil containing a mixture of saturated and unsaturated hydrocarbons? Explain your reasoning.
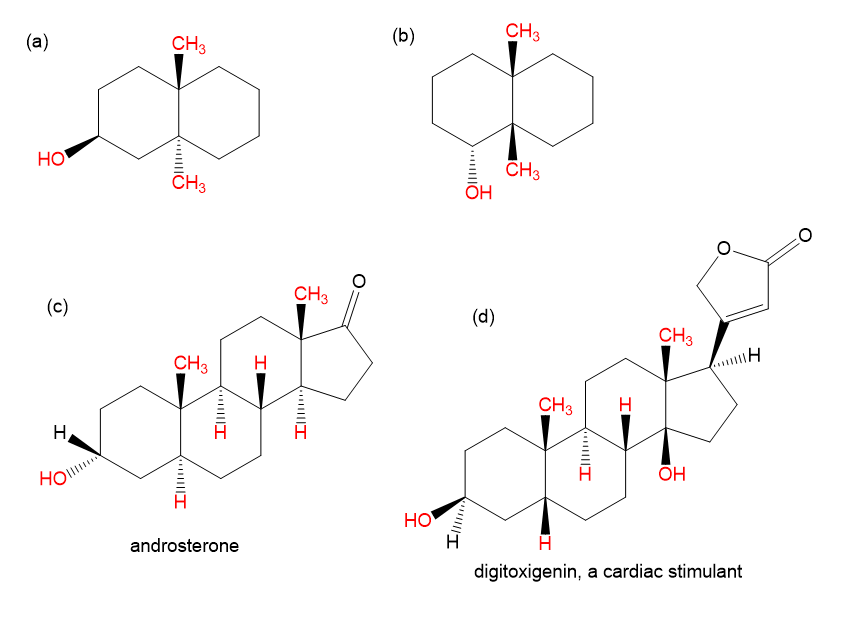
Draw each molecule in a stable chair conformation and indicate whether each red group is axial or equatorial.
What do you think about this solution?
We value your feedback to improve our textbook solutions.