Chapter 25: Q10P (page 1314)
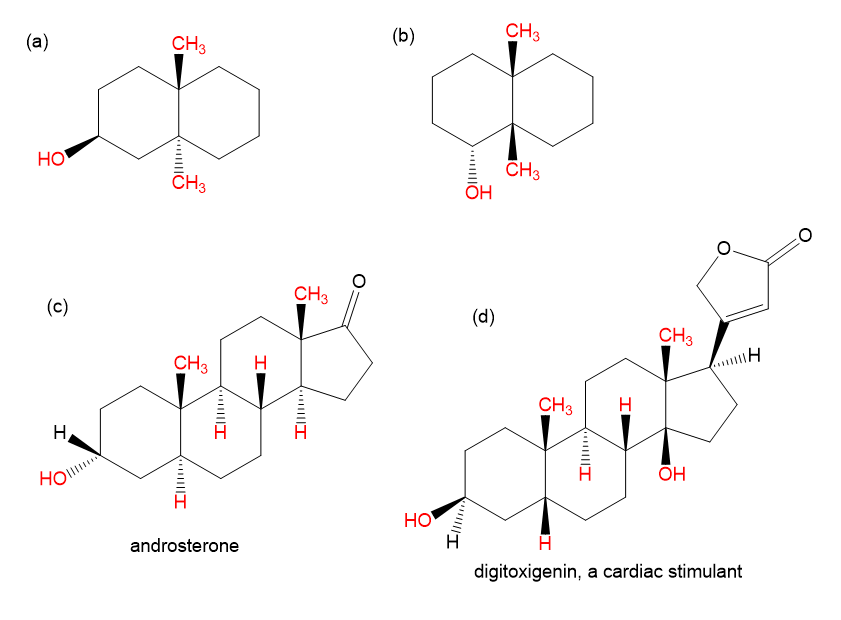
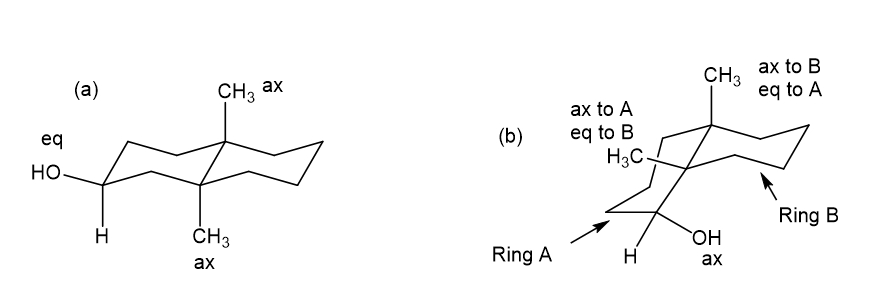
Draw each molecule in a stable chair conformation and indicate whether each red group is axial or equatorial.
Short Answer
Learning Materials
Features
Discover
Chapter 25: Q10P (page 1314)
Draw each molecule in a stable chair conformation and indicate whether each red group is axial or equatorial.



All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of an optically active triglyceride containing one equivalent of stearic acid and two equivalents of oleic acid. Draw the products expected when this triglyceride reacts with the following reagents. In each case, predict whether the products will be optically active.
Question: Trimyristin, a solid fat present in nutmeg, is hydrolyzed to give one equivalent of glyceroland three equivalents of myristic acid. Give the structure of trimyristin.
Give an equation for the complete hydrogenation of trilinolein using an excess of hydrogen. Name the product and predict approximate melting points for the starting material and the product.
Phospholipids undergo saponification much like triglycerides. Draw the structure of a phospholipid meeting the following criteria. Then draw the products that would result from its saponification.
(a) a cephalin containing stearic acid and oleic acid
(b) a lecithin containing palmitic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.