Chapter 18: Q20. (page 945)
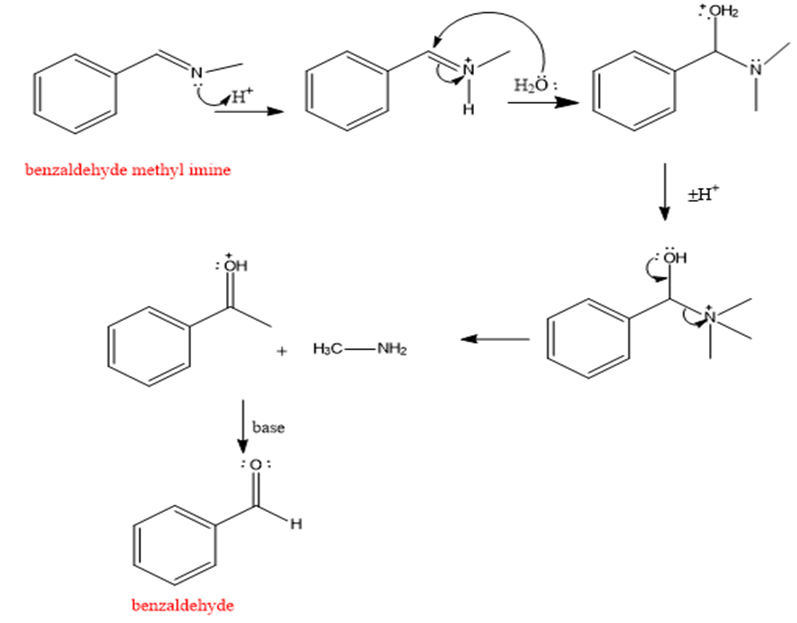
Propose a mechanism for the hydrolysis of benzaldehyde methyl imine just shown.
Short Answer
The mechanism for hydrolysis is shown below.
Learning Materials
Features
Discover
Chapter 18: Q20. (page 945)
Propose a mechanism for the hydrolysis of benzaldehyde methyl imine just shown.
The mechanism for hydrolysis is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
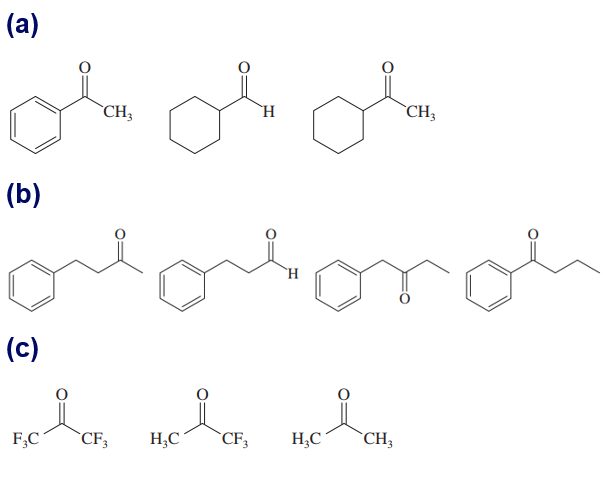
Question: Within each set of structures, indicate which will react fastest, and which slowest, toward nucleophilic addition in basic conditions.
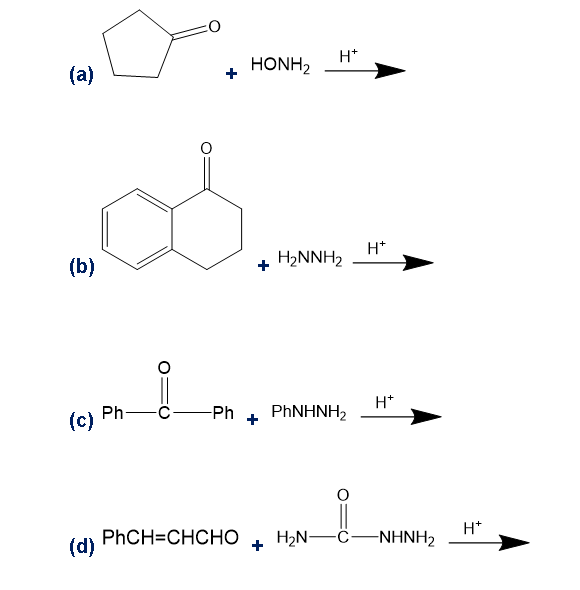
Predict the products of the following reactions.
Sketch the expected proton NMR spectra of 3,3- dimethylbutanal.
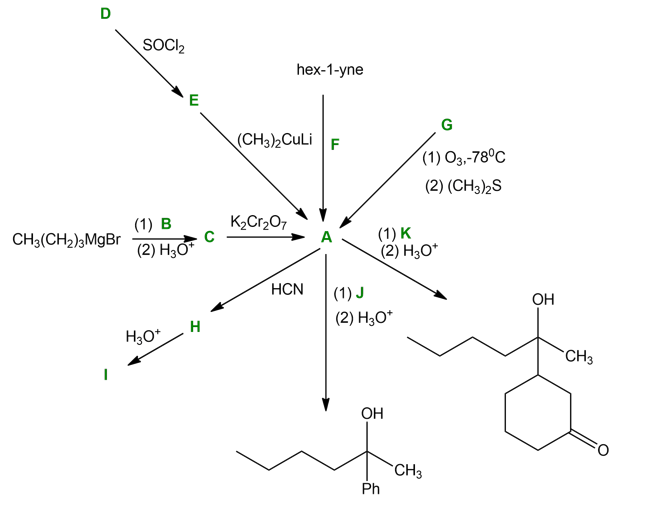
Question: Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K.
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one?
What do you think about this solution?
We value your feedback to improve our textbook solutions.