Chapter 14: Q2P. (page 712)
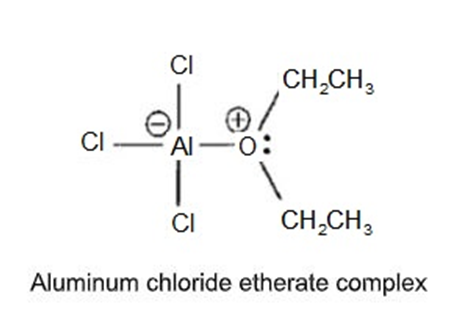
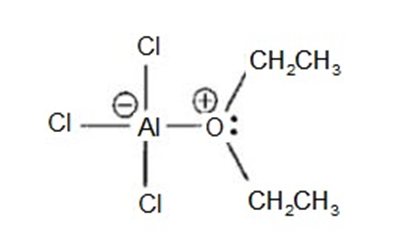
Aluminum trichloride () dissolves in ether with the evolution of a largeamount of heat. (In fact, this reaction can become rather violent if it gets too warm.)Show the structure of the resulting aluminum chloride etherate complex.
Short Answer