Chapter 14: Q22P (page 733)
Propose mechanisms for the epoxidation and ring-opening steps of the epoxidation and hydrolysis of  shown above. Predict the product of the same reaction with.
shown above. Predict the product of the same reaction with.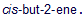
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 14: Q22P (page 733)
Propose mechanisms for the epoxidation and ring-opening steps of the epoxidation and hydrolysis of  shown above. Predict the product of the same reaction with.
shown above. Predict the product of the same reaction with.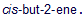
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish the following transformations. Some of these examples require more than one step.

Aluminum trichloride () dissolves in ether with the evolution of a largeamount of heat. (In fact, this reaction can become rather violent if it gets too warm.)Show the structure of the resulting aluminum chloride etherate complex.
Write structural formulas for the following compounds
(d) diallyl ether (e) allyl ethyl ether (f) cycloheptane oxide
(g) trans-2,3-epoxyheptane (h) (2R,3S)-2-ethoxypentan-3-ol (i) cis-2,3-dimethyloxirane
In 2012, a group led by Professor Masayuki Satake of the University of Tokyo reported the isolation and structure determination of a toxin from a marine algal bloom that decimated the fish population off the New Zealand coast in 1998. Extensive mass spectrometry and NMR experiments ultimately led to the structure shown below, named Brevisulcenal-F. (See Journal of the American Chemical Society, 2012, 134, 4963-4968.) This structure holds the record for the largest number of fused rings, at 17.
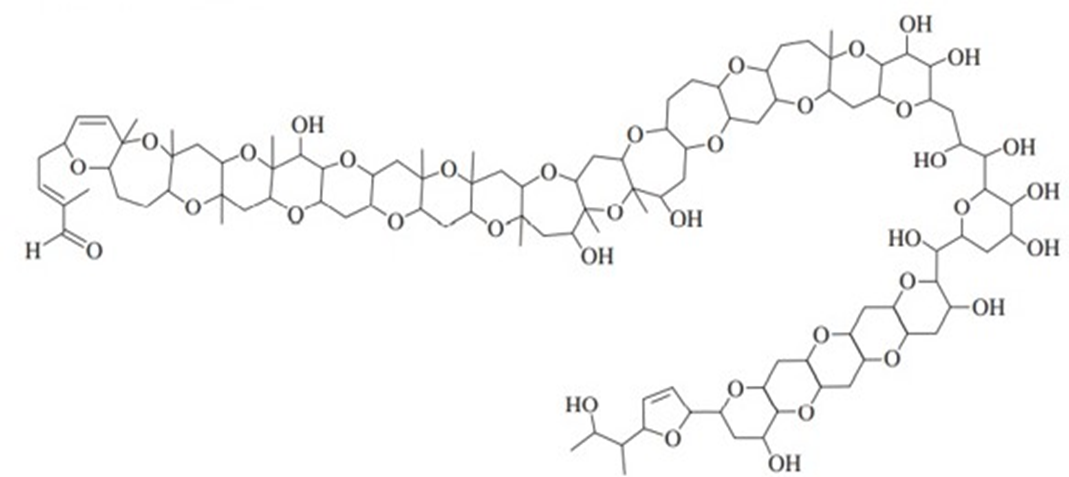
Brevisulcenal-F
a. How many ether groups are present?
b. How many alcohol groups are present? Classify the alcohols as 1o or 2o or 3o.
c. Are there any other oxygen-containing functional groups? Which, if any?
Question. Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism for the formation of propene and explain why it is favored at higher temperatures.
What do you think about this solution?
We value your feedback to improve our textbook solutions.