Chapter 15: Q 15P (page 772)
What dienes and dienophiles would react to give the following Diels-Alder products?
a)

b)
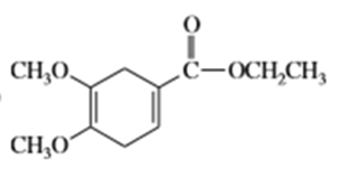
c)
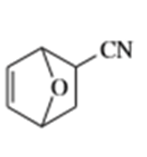
d)
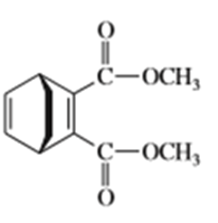
e)

f)
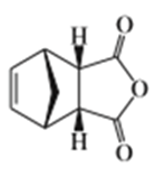
Short Answer
a)
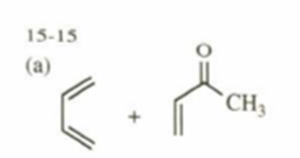
b)
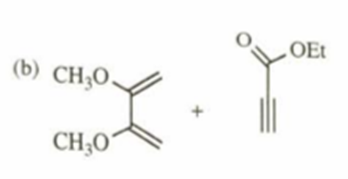
c)
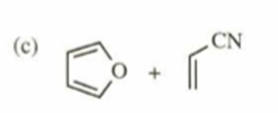
d)

e)
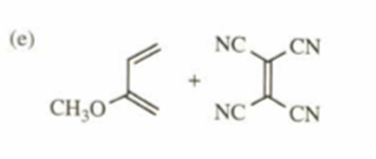
f)

Learning Materials
Features
Discover
Chapter 15: Q 15P (page 772)
What dienes and dienophiles would react to give the following Diels-Alder products?
a)

b)

c)

d)

e)

f)

a)

b)

c)

d)

e)

f)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: When is added to buta-1,3- diene at -150C, the product mixture contains 60% of product A and 40% of product B. When the same reaction takes place at 600C, the product ratio is 10% A and 90% B.
a. Propose structures for products A and B (Hint: In many case, an allylic carbocation is more stable than a bromonium ion.)
b. Propose a mechanism to account for formation of both A and B.
c. Show why A predominates at -150C and B predominates at 600C .
d. If you had a solution of pure A, and its temperature were raised to 600C , what would you expect to happen ? Propose a mechanism to support your prediction.
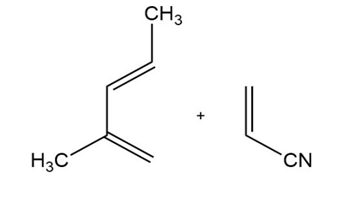
Predict the products of the following Diels-Alder reactions.
a)
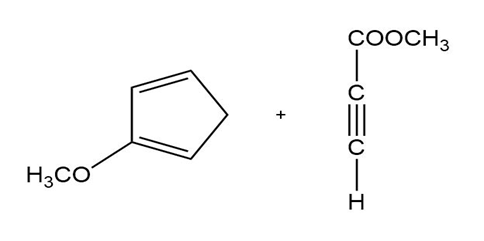
b)

c)

d)
When methylenecyclohexane is treated with a low concentration of bromine under irradiation by a sunlamp, two substitution products are formed.

One milligram of a compound of molecular weight 160 is dissolved in 10 mL of ethanol, and the solution is poured into a 1-cm UV cell. The UV spectrum is taken, and there is an absorption at 𝛌max= 247 nm.The maximum absorbance at 247 nm is 0.50. Calculate the value of𝛆 for this absorption.
Show how you might synthesize the following compounds starting with bromobenzene, and alkyl or alkenyl halides of four carbon atoms or fewer.
a. 3-phenylprop-1-ene
b.5-methylhex-2-ene
c. dec-5-ene
What do you think about this solution?
We value your feedback to improve our textbook solutions.