Chapter 15: Q 18P (page 778)
Predict the products of the following Diels-Alder reactions.
a)
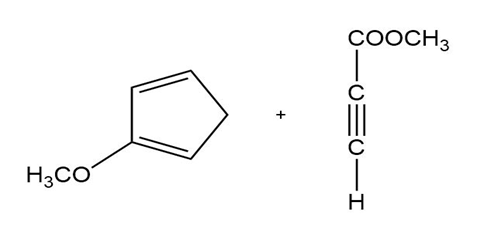
b)
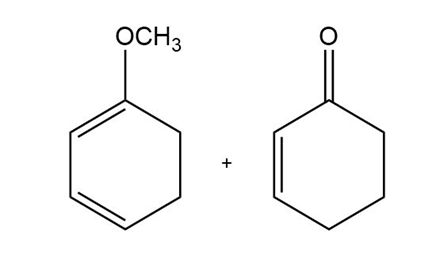
c)
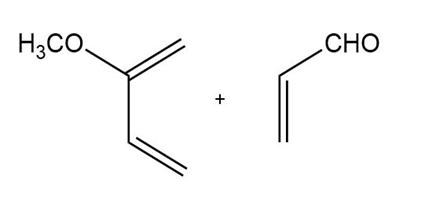
d)
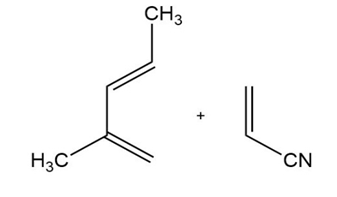
Short Answer
a)
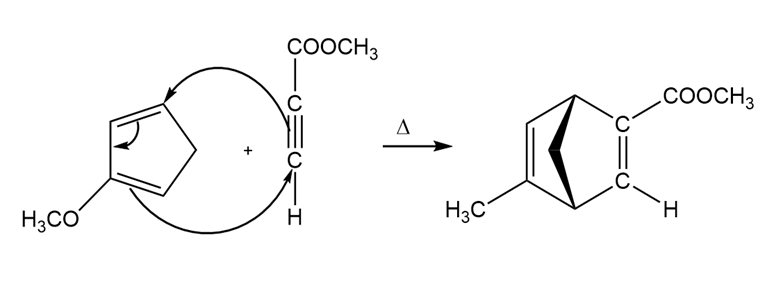
b)
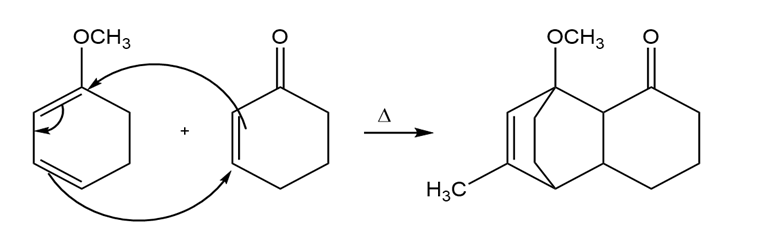
c)
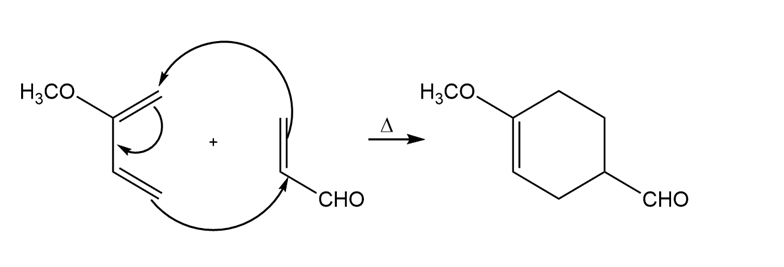
d)
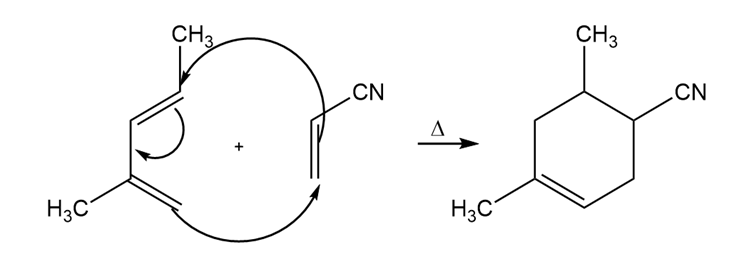
Learning Materials
Features
Discover
Chapter 15: Q 18P (page 778)
Predict the products of the following Diels-Alder reactions.
a)

b)

c)

d)

a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: When Br2 is added to buta-1,3- diene at -150C , the product mixture contains 60% of product A and 40% of product B. When the same reaction takes place at 600C , the product ratio is 10% A and 90% B.
a. Propose structures for products A and B (Hint: In many case, an allylic carbocation is more stable than a bromonium ion.)
b. Propose a mechanism to account for formation of both A and B.
c. Show why A predominates at -150C and B predominates at 600C .
d. If you had a solution of pure A, and its temperature were raised to600C , what would you expect to happen ? Propose a mechanism to support your prediction.
Question: Draw another resonance form for each of the substituted allylic cations shown in the preceding figure, showing how the positive charge is shared by another carbon atom. In each case, state whether your second resonance form is a more important or less important resonance contributor than the first structure. (Which structure places the positive charge on the more-substituted carbon atom?)
When methylenecyclohexane is treated with a low concentration of bromine under irradiation by a sunlamp, two substitution products are formed.
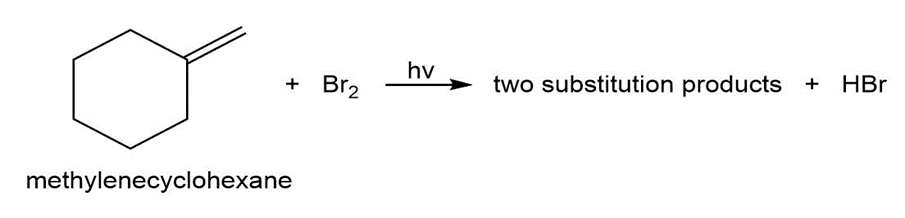
Question: When N-bromosuccinimide is added to hex-1-ene in and a sunlamp is shone on the mixture, three products result.
Question: When 3-bromo-1-methylcyclohexene undergoes solvolysis in hot ethanol, two products are formed. Propose a mechanism that accounts for both of these products.
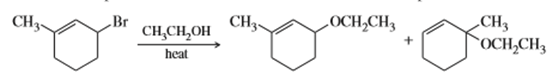
What do you think about this solution?
We value your feedback to improve our textbook solutions.