Chapter 15: Q 13P (page 770)
Show how you might synthesize the following compounds starting with bromobenzene, and alkyl or alkenyl halides of four carbon atoms or fewer.
a. 3-phenylprop-1-ene
b.5-methylhex-2-ene
c. dec-5-ene
Short Answer
a)
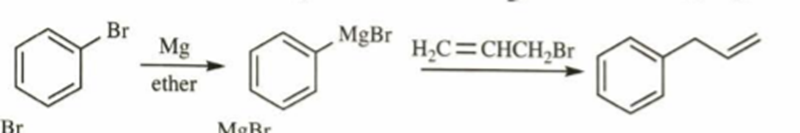
b)
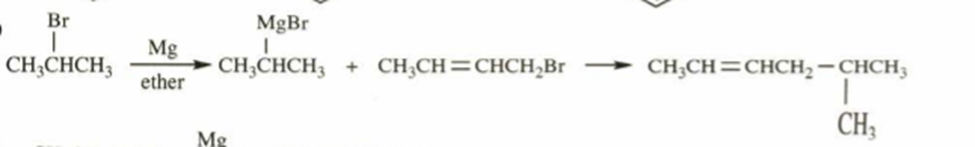
c)
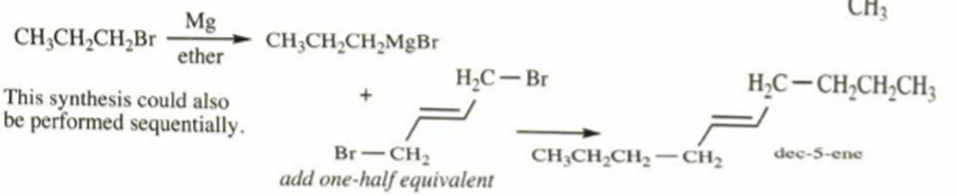
Learning Materials
Features
Discover
Chapter 15: Q 13P (page 770)
Show how you might synthesize the following compounds starting with bromobenzene, and alkyl or alkenyl halides of four carbon atoms or fewer.
a. 3-phenylprop-1-ene
b.5-methylhex-2-ene
c. dec-5-ene
a)
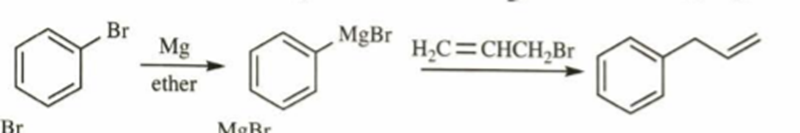
b)
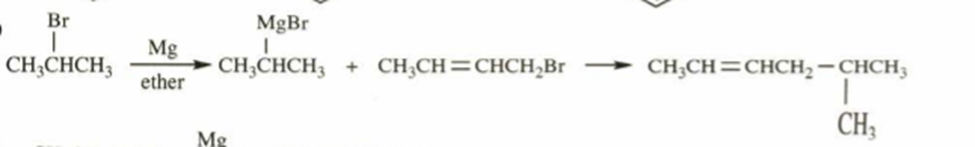
c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Treatment of an alkyl halide with AgNO3 in alcohol often promotes ionization.
Ag+ + R-Cl → AgCl + R+
When 4-chloro-2-methylhex-2-ene reacts with AgNO3 in ethanol, two isomeric ethers are formed. Suggest structures, and propose a mechanism for their formation.
(a)Show that the [4+4] cycloaddition of two butadiene molecules to give cycloocta-1,5-diene is thermally forbidden but photochemically allowed.
(b)There is a different, thermally allowed cycloaddition of two butadiene molecules. Show this reaction, and explain why it is thermally allowed. (Hint: Consider the dimerization of cyclopentadiene.)
What dienes and dienophiles would react to give the following Diels-Alder products?
a)
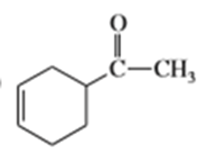
b)
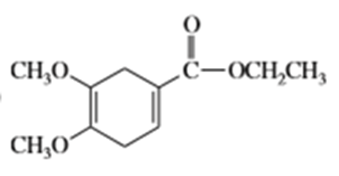
c)
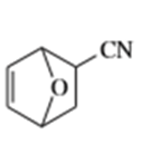
d)
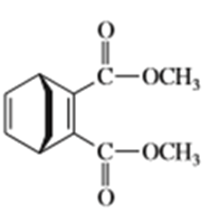
e)
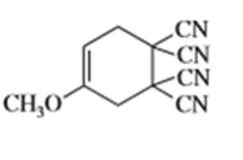
f)
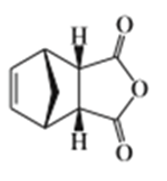
Predict the products of the following Diels-Alder reactions.
a)
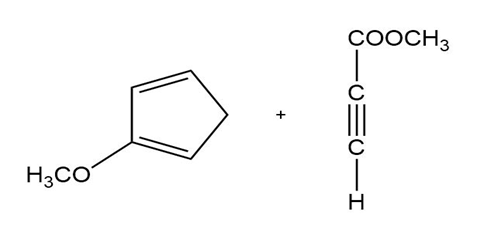
b)
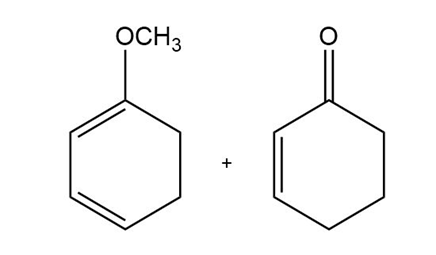
c)
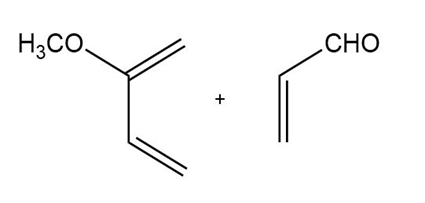
d)
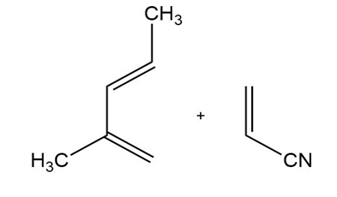
Question: When Br2 is added to buta-1,3- diene at -150C , the product mixture contains 60% of product A and 40% of product B. When the same reaction takes place at 600C , the product ratio is 10% A and 90% B.
a. Propose structures for products A and B (Hint: In many case, an allylic carbocation is more stable than a bromonium ion.)
b. Propose a mechanism to account for formation of both A and B.
c. Show why A predominates at -150C and B predominates at 600C .
d. If you had a solution of pure A, and its temperature were raised to600C , what would you expect to happen ? Propose a mechanism to support your prediction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.