Chapter 22: Q18P (page 1166)
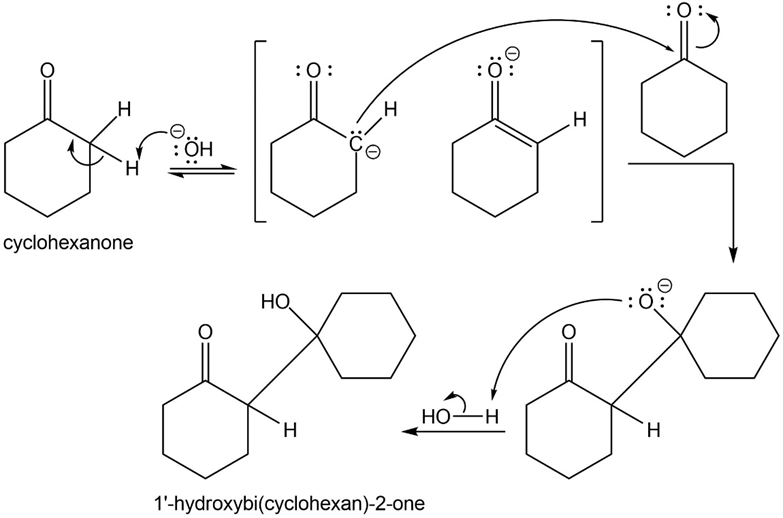
Propose a mechanism for the aldol condensation of cyclohexanone. Do you expect the equilibrium to favor the reactant or the product?
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q18P (page 1166)
Propose a mechanism for the aldol condensation of cyclohexanone. Do you expect the equilibrium to favor the reactant or the product?

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how an acetoacetic ester synthesis might be used to form a δ -diketone such as heptane-2,6-dione.
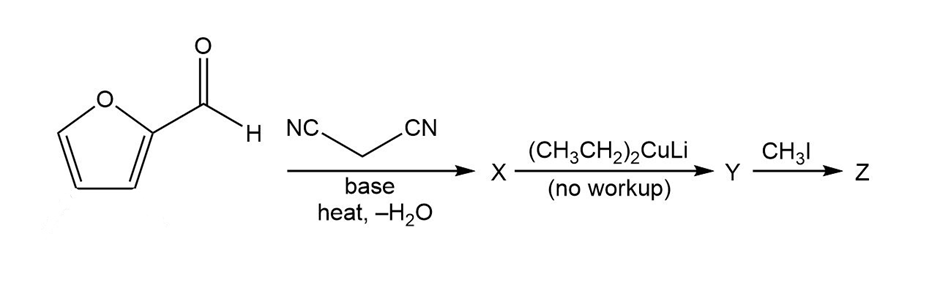
Predict the products from this sequence of reactions
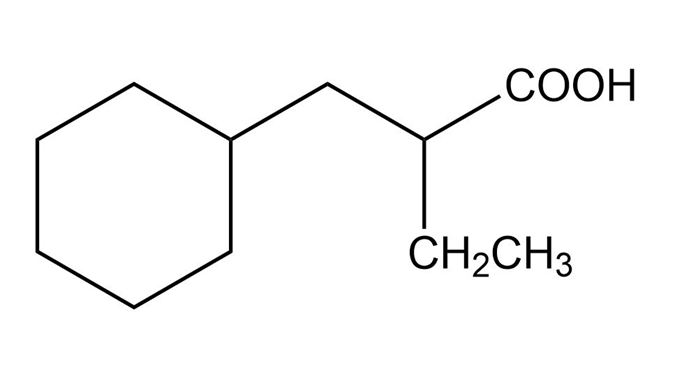
Show how you would use the malonic ester synthesis to make the following compounds
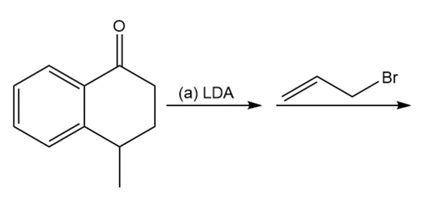
Question: Predict the products of these reaction sequences.
a.
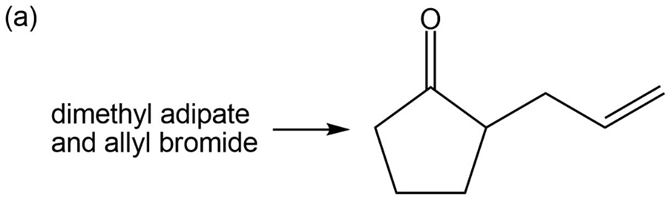
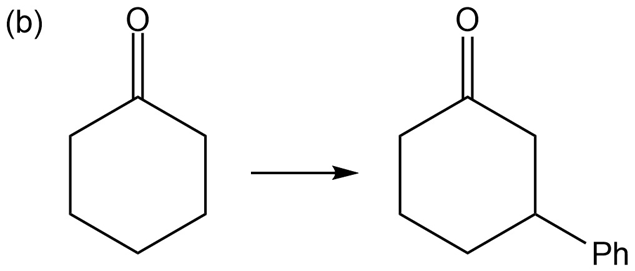
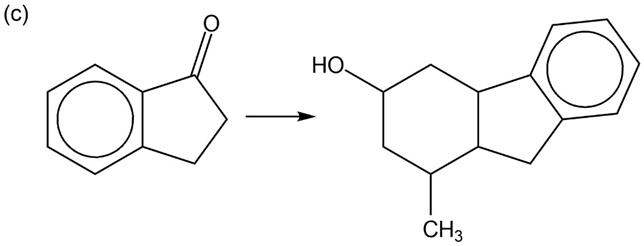
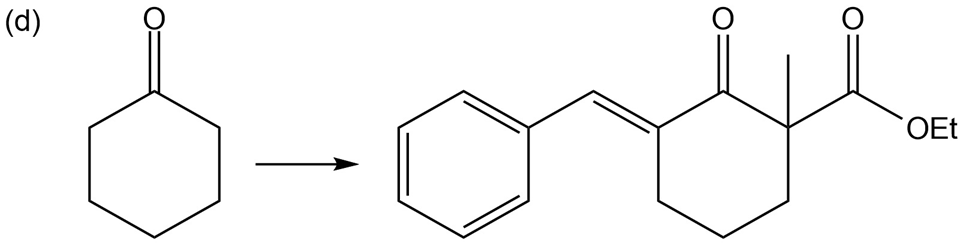
Show how you would accomplish the following multistep conversions. You may use any additional reagents you need.
What do you think about this solution?
We value your feedback to improve our textbook solutions.