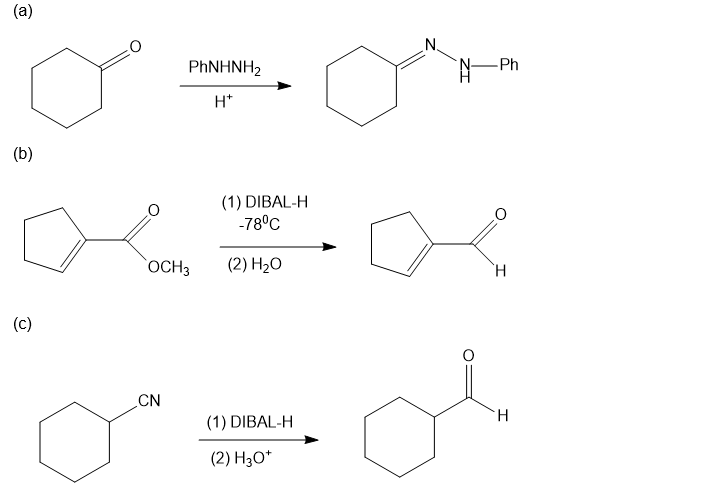
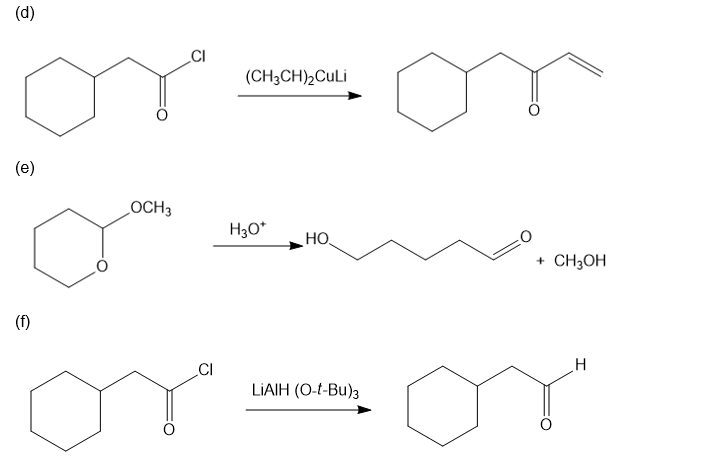
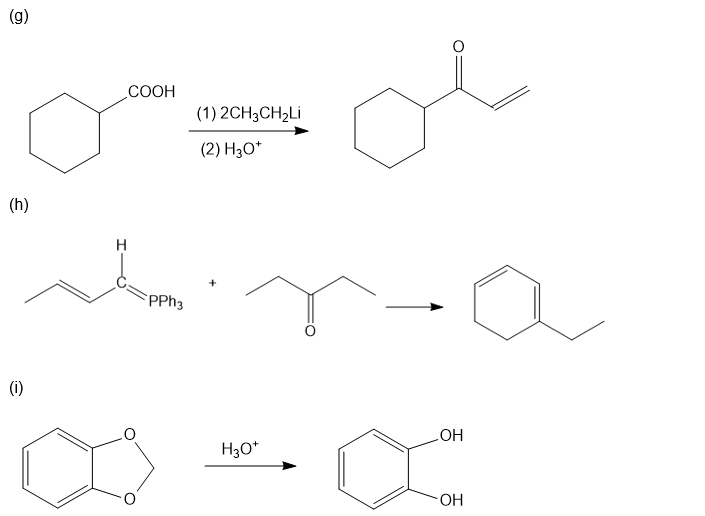
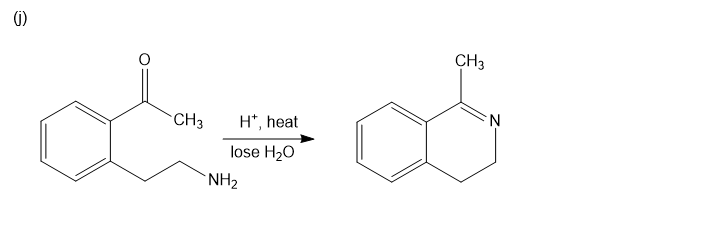
Chapter 20: Q39P (page 1038)
Predict the major products of the following reactions.
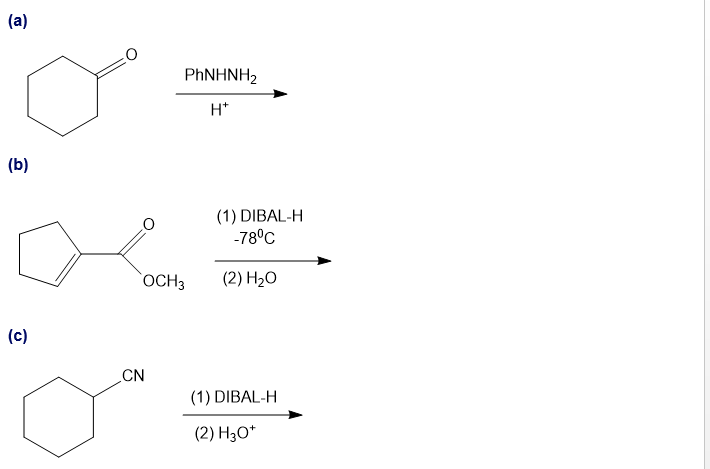
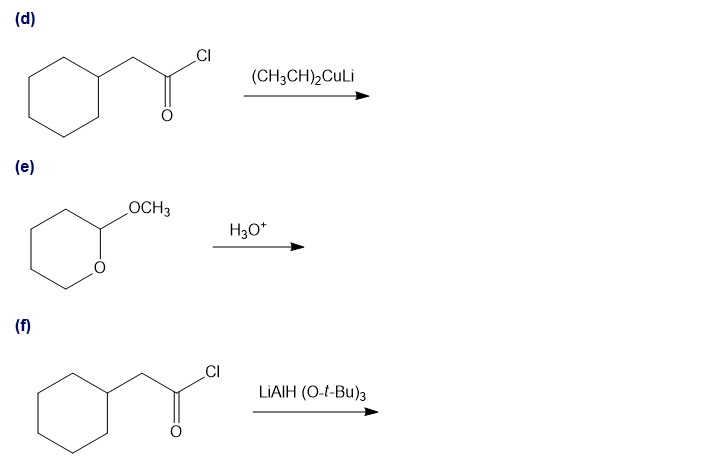
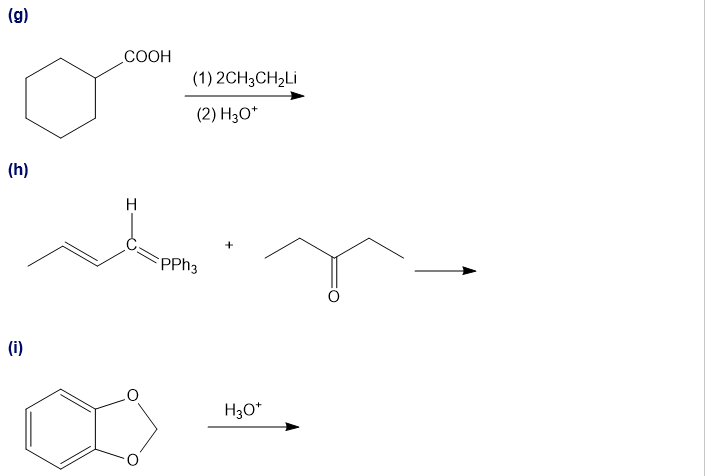
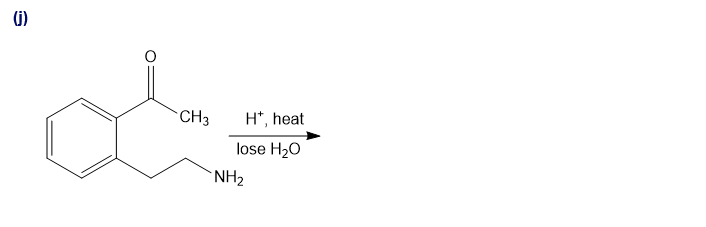
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q39P (page 1038)
Predict the major products of the following reactions.








All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish the following multistep syntheses using the indicated starting material and any necessary reagents.
(a) hept-6-en-1-ol caprolactone
(b) methoxybenzenep-methoxybenzamide
(c)
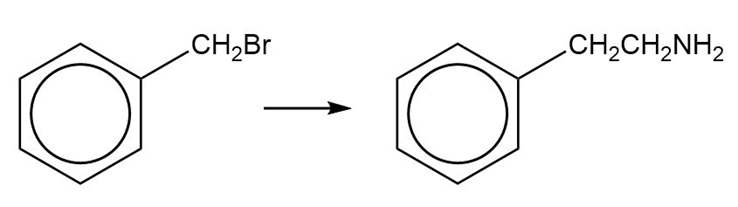
(d)
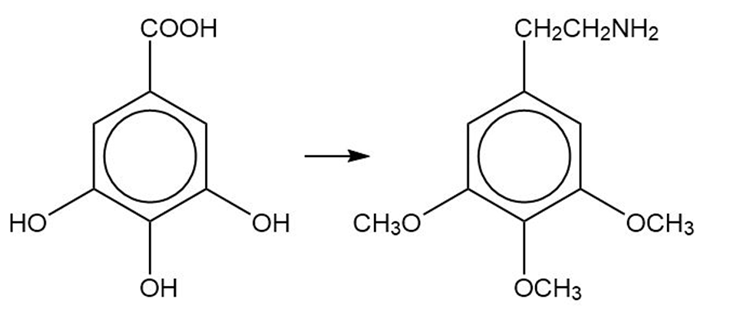
Gallic acid Mescaline
Question: Predict the products (if any) of the following acid-base reactions.
Q. 15 Show how Fischer esterification might be used to form the following esters. In each case, suggest a method for driving the reaction to completion.
Suggest how you would convert trans-4-methylcyclohexanol to
(a) trans-1-chloro-4-methylcyclohexane.
(b) cis-1-chloro-4-methylcyclohexane.
The following reaction takes place under second-order conditions (strong nucleophile), yet the structure of the product shows rearrangement. Also, the rate of this reaction is several thousand times faster than the rate of substitution of hydroxide ion on 2-chlorobutane under similar conditions. Propose a mechanism to explain the enhanced rate and rearrangement observed in this unusual reaction. (“Et” is the abbreviation for ethyl.
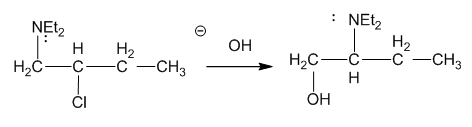
What do you think about this solution?
We value your feedback to improve our textbook solutions.