Chapter 20: Q19P (page 1038)
Suggest how you would convert trans-4-methylcyclohexanol to
(a) trans-1-chloro-4-methylcyclohexane.
(b) cis-1-chloro-4-methylcyclohexane.
Short Answer
(a)
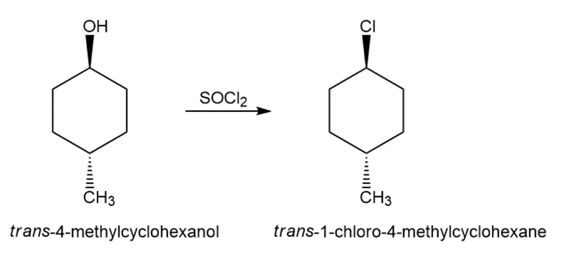
(b)
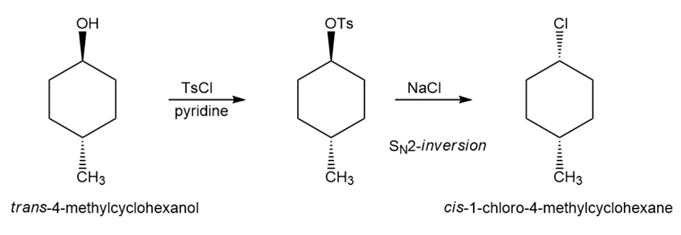
Learning Materials
Features
Discover
Chapter 20: Q19P (page 1038)
Suggest how you would convert trans-4-methylcyclohexanol to
(a) trans-1-chloro-4-methylcyclohexane.
(b) cis-1-chloro-4-methylcyclohexane.
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
In the presence of 18-crown-6, potassium permanganate dissolves inbenzene to give “purple benzene” a useful reagent for oxidizing alkenes in an aprotic environment. Use a drawing of the complex to show why dissolves in benzeneand why the reactivity of the permanganate ion is enhanced.
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
(a)Hydrogen peroxide (HOOH) has a pKa of 11.6, making it roughly 10,000 times as strong an acid as water (pKa = 15.7). Explain why H2O2is a stronger acid than H2O.
(b) In contrast to part (a), peroxyacetic acid (pKa = 8.2) is a much weakeracid than acetic acid (pka = 4.74) . Explain why peroxyacetic acid is a weaker acid than acetic acid.
(c) Peroxyacetic acid (bp = 105° C) has a lower boiling point than acetic acid (bp = 118° C) , even though peroxyacetic acid has a higher molecular weight. Explain why peroxyacetic acid is more volatile than acetic acid.
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)
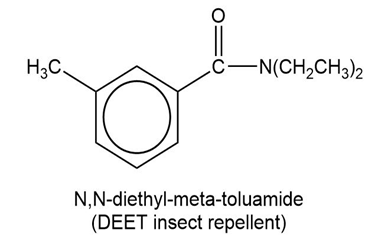
(b)
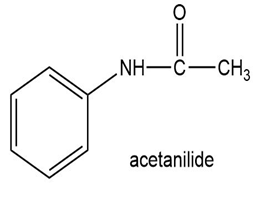
(c)
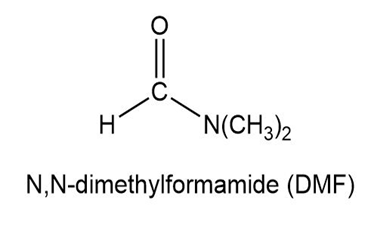
Circle the isoprene units in geranial, menthol, camphor, and abietic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.