a. Isoxazole is an aromatic compound because it has4 pielectrons, and one of the lone pairs of oxygen participates in an aromatic sextet. So, it obeys Huckel’s rule.
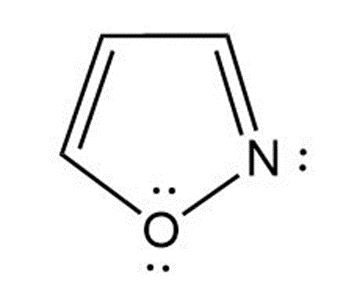
![]()
Isoxazole
(Aromatic)
b. 1,3-thiazole is an aromatic compound because it has 4 pi-electrons and one of the lone pairs of sulfur participates in the aromatic sextet. So, it obeys Huckel’s rule.
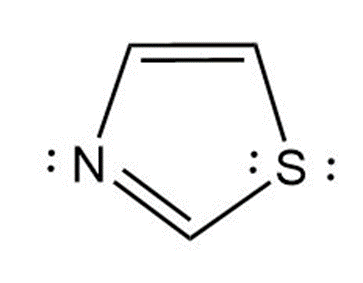
1,3-thiazole
(Aromatic)
c. Pyran is a nonaromatic compound because it has no conjugated pi-electron system. So, it does not obey Huckel’s rule.
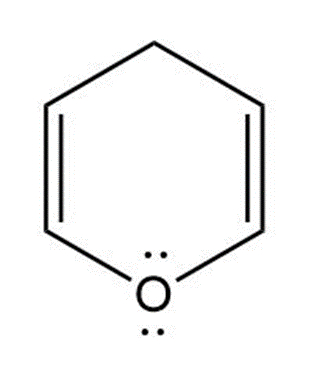
Pyran
(Nonaromatic)
d. Pyrylium ion is an aromatic compound because it has 4pi-electrons and one of the lone pairs of oxygen participates in an aromatic sextet. Here, the carbocation also undergoes resonance. So, it obeys Huckel’s rule.
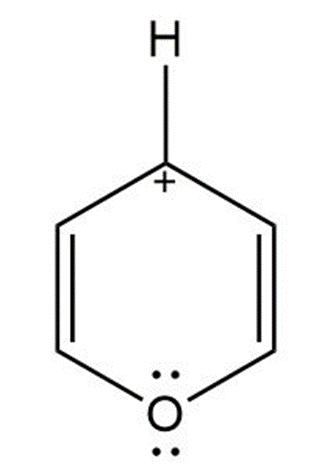
Pyrylium ion
(Aromatic)
e. Pyrone is an aromatic compound because it has 4-pi electrons and one of the lone pairs of oxygen participates in forming an aromatic sextet. So, it obeys Huckel’s rule.
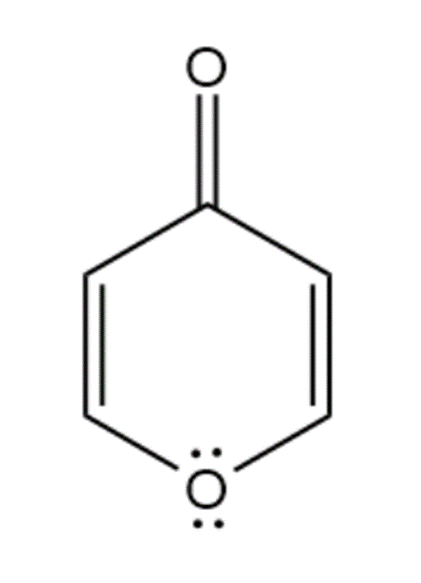
Pyrone
(Aromatic)
f. 1,2-Dihydropyridine is a nonaromatic compound because it has no conjugated pi electron system and it contains one sp3 hybridized carbon atom. So, it does not obey Huckel’s rule.
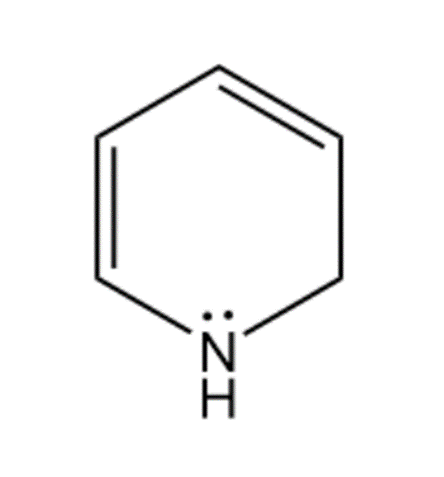
1,2-Dihydropyridine
(Nonaromatic)
g. Cytosine is aromatic because it has 4pi- electrons and one lone pair of nitrogen participates in the formation of an aromatic sextet. So, it obeys Huckel’s rule.
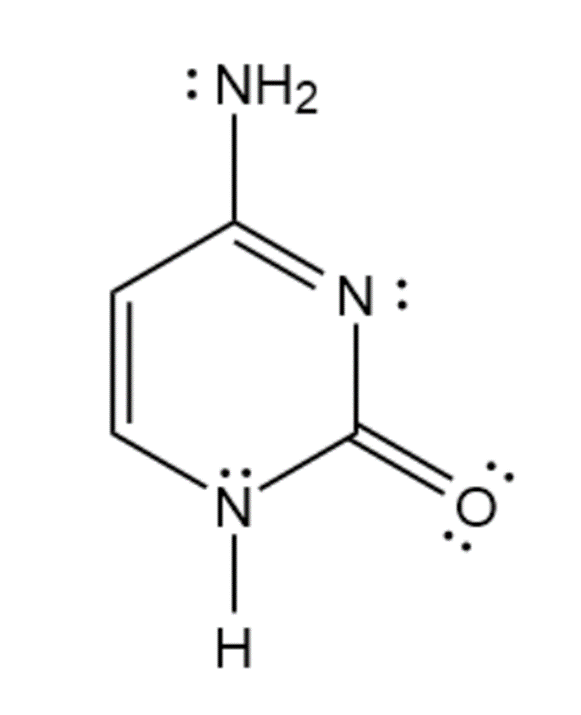
Cytosine
(Aromatic)
h. The given compound is antiaromatic as it contains 8pi-electrons through the 4n pi-electron rule.
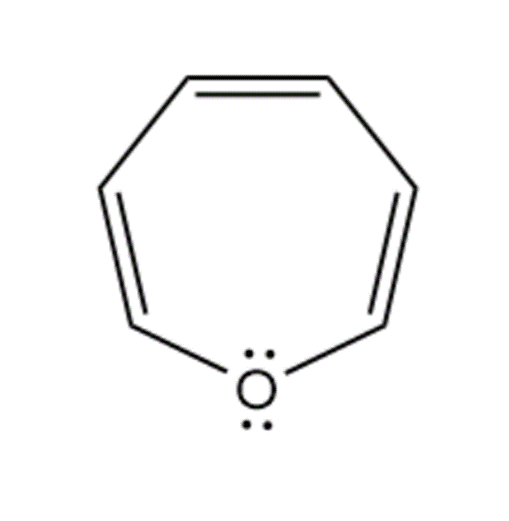
Antiaromatic




























