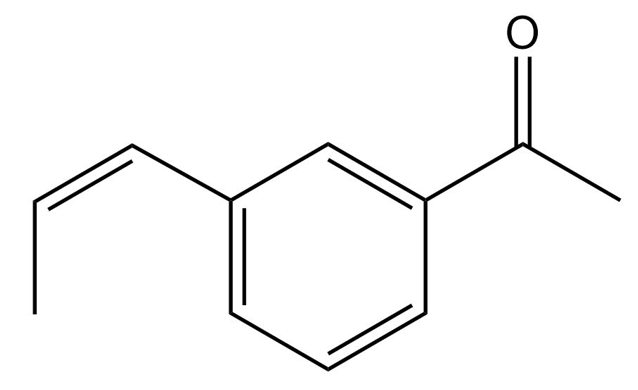
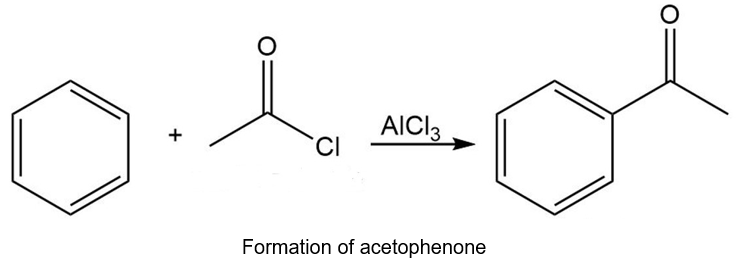
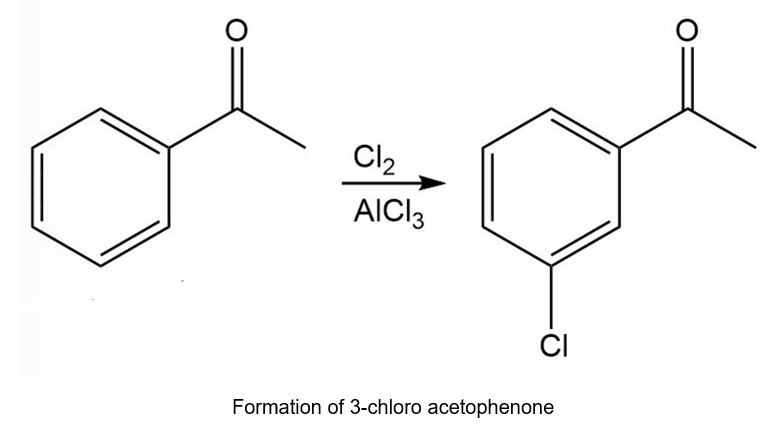
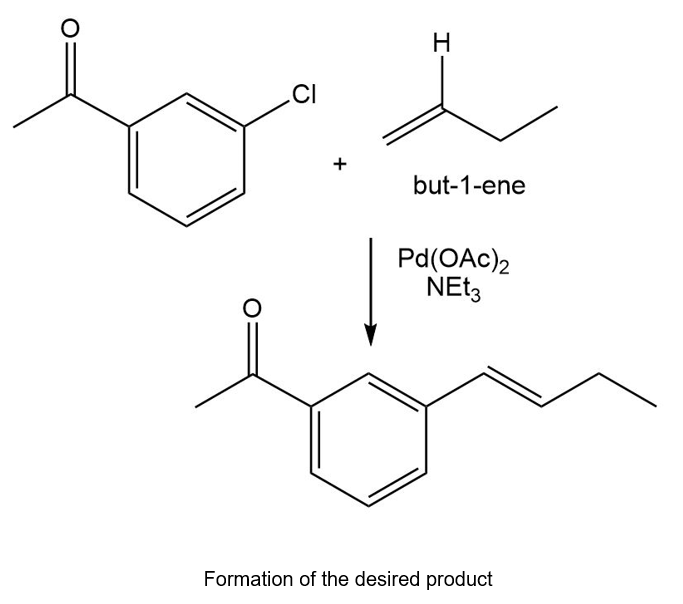
Chapter 20: 72P (page 1038)
Starting with benzene and any other reagents you need, show how you would synthesize the compound shown here. (Hint: Consider a Pd-catalyzed coupling for the final step).
Short Answer
Learning Materials
Features
Discover
Chapter 20: 72P (page 1038)
Starting with benzene and any other reagents you need, show how you would synthesize the compound shown here. (Hint: Consider a Pd-catalyzed coupling for the final step).




All the tools & learning materials you need for study success - in one app.
Get started for free
Show how Fischer esterification might be used to form the following esters. In each case, suggest a method for driving the reaction to completion.
(a) methyl salicylate
(b) methyl formate (bp 32C)
(c) ethyl phenylacetate
For each compound,
(a)
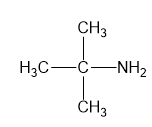
(b)
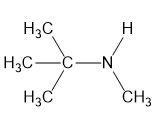
(c)
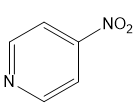
(d)
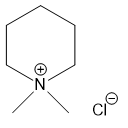
(e)
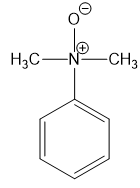
(f)
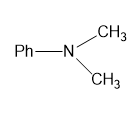
(g)
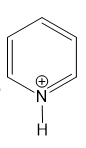
(h)
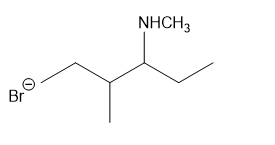
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)
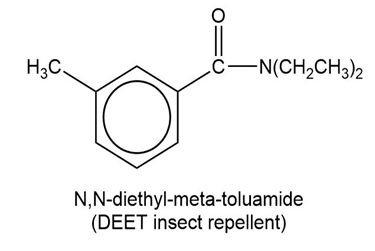
(b)
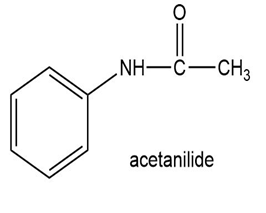
(c)

Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
(a)
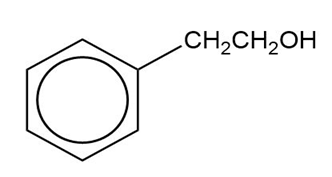
(b)

(c)
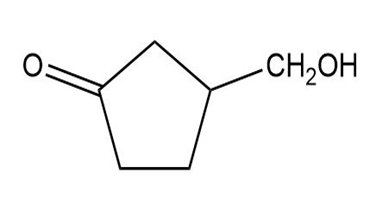
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.