Chapter 21: Q36P-b (page 1129)
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Short Answer
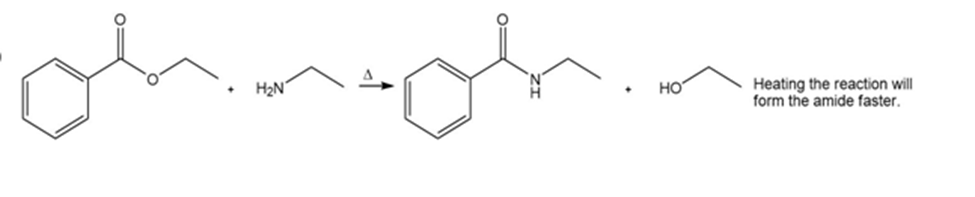
Synthetic transformation for ethyl benzoate to N-ethylbenzamide is shown as,
Learning Materials
Features
Discover
Chapter 21: Q36P-b (page 1129)
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Synthetic transformation for ethyl benzoate to N-ethylbenzamide is shown as,

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the formation of 9-hydroxynonanoic acid lactone, as show in the preceding figure.
Show how you would accomplish the following syntheses using amides as intermediates.
You may use any necessary reagents.
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Show how you would accomplish the following syntheses using amides as intermediates.
You may use any necessary reagents.
Amides, ketones, carboxylic acids, and aldehydes all give strong carbonyl stretching absorptions in the IR spectrum. How can you use other peaks in their IR spectra to distinguish among these four common functional groups?
What do you think about this solution?
We value your feedback to improve our textbook solutions.