Chapter 21: Q36P-a (page 1129)
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Short Answer
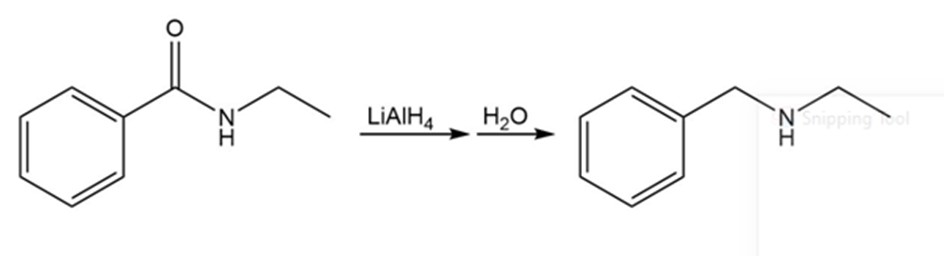
Synthetic transformation for N-ethylbenzamide to benzylethylamine is show as,
Learning Materials
Features
Discover
Chapter 21: Q36P-a (page 1129)
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Synthetic transformation for N-ethylbenzamide to benzylethylamine is show as,

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use appropriate acyl chlorides and amines to synthesize the following amides.
(a)N,N-dimethylacetamide
(b)acetanilide
(c)cyclohexanecarboxamide
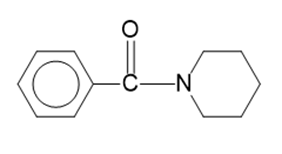
(d)
Show how you would use anhydrides to synthesize the following compounds. In each case, explain why an anhydride might be preferable to an acid chloride.
(c) phthalic acid monoamide
Show how each transformation may be accomplished by using a nitrile as an intermediate.
You may use any necessary reagents.
(a)Propose a mechanism for the reaction of benzyl alcohol with acetyl chloride to give benzyl acetate.
(b) Propose a mechanism for the reaction of benzoic acid with acetyl chloride to give acetic benzoic anhydride.
(c)Propose a secondmechanism for the reaction of benzoic acid with acetyl chloride to give acetic benzoic anhydride. This time, let the otheroxygen of benzoic acid serve as the nucleophile to attack the carbonyl group of acetyl chloride. Because proton transfers are fast between these oxygen atoms, it is difficult to differentiate between these two mechanisms experimentally.
(d)Propose a mechanism for the reaction of aniline with acetic anhydride to give acetanilide.
(e)Propose a mechanism for the reaction of aniline with ethyl acetate to give acetanilide. What is the leaving group in your proposed mechanism? Would this be a suitable leaving group for an reaction?
One mole of acetyl chloride is added to a liter of triethylamine, resulting in a vigorous exothermic reaction. Once the reaction mixture has cooled, 1 mole of ethanol is added. Another vigorous exothermic reaction results. The mixture is analyzed and found to contain triethylamine, ethyl acetate, and triethylammonium chloride. Propose mechanisms for the two exothermic reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.