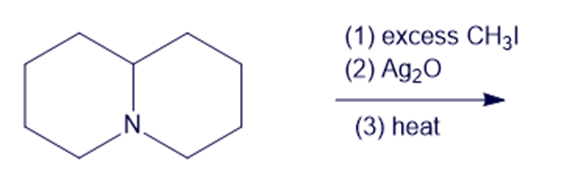
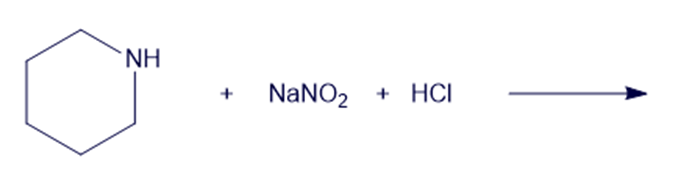
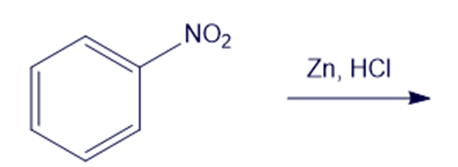
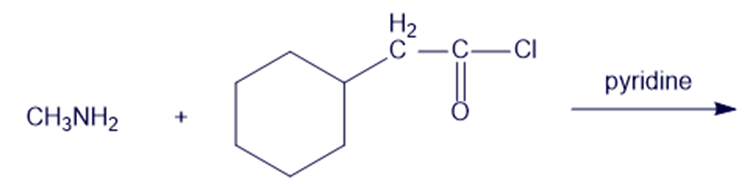
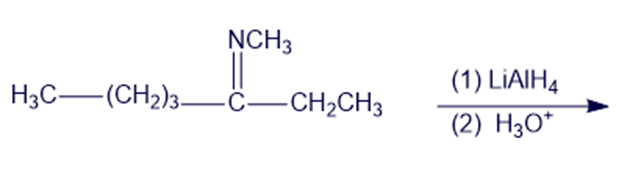
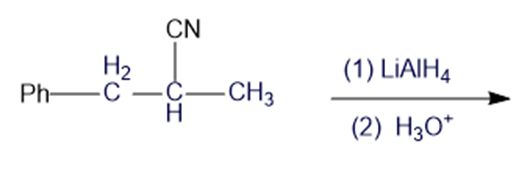
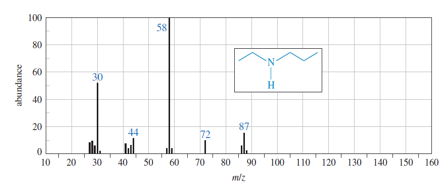
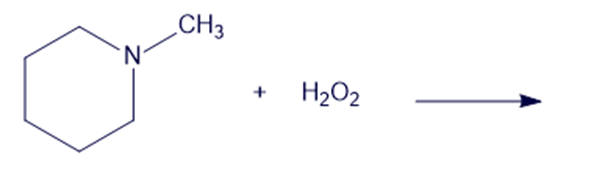Chapter 19: Q6P (page 988)
Rank each set of compounds in order of increasing basicity
(a) NaOH, NH3, CH3NH2, Ph-NH2
(b) aniline, p-methylaniline, p-nitroaniline
(c) aniline, pyrrole, pyridine, piperidine
(d) pyrrole, imidazole, 3-nitropyrrole
Short Answer
(a)
Basicity (pKb) of NaOH is 0.2
Basicity (pKb) of NH3 is 4.74
Basicity (pKb) of CH3NH2 is 3.38
Basicity (pKb) of Ph-NH2 is 9.38
Order of increasing basicity = Ph-NH2 < NH3 <CH3NH2 < NaOH
(b)
Basicity (pKb) of aniline is 9.38
Basicity (pKb) of p-methylaniline is 9.21
Basicity (pKb) of p-nitroaniline is 12.99
Order of increasing basicity = p-nitroaniline< aniline < p-nitroaniline
(c)
Basicity (pKb) of aniline is 9.38
Basicity (pKb) of pyrrole is 13.6
Basicity (pKb) of pyridine is 8.77
Basicity (pKb) of piperidine is 4
Order of increasing basicity = pyrrole < aniline < pyridine < piperidine
(d)
Basicity (pKb) of pyrrole is 13.6
Basicity (pKb) of imidazole is 7.0
3-nitropyrrole is formed by adding nitric acid to pyrrole i.e. nitration of pyrrole group.
Therefore it is less basic than pyrrole.
Order of increasing basicity = 3-nitopyrrole< pyrrole < imidazole