Chapter 19: Q27P (page 1020)
Show how to synthesize the following amines from the indicated starting materials by acylation–reduction.
(a) N-butylpiperidine from piperidine
(b) N-benzylaniline from anilinea
Short Answer
(a)
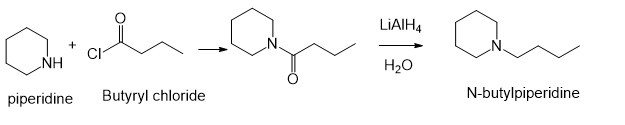
(b)
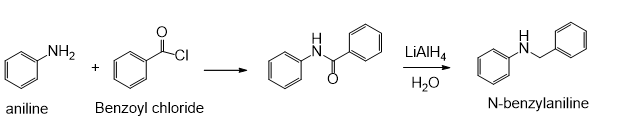
Learning Materials
Features
Discover
Chapter 19: Q27P (page 1020)
Show how to synthesize the following amines from the indicated starting materials by acylation–reduction.
(a) N-butylpiperidine from piperidine
(b) N-benzylaniline from anilinea
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
a) Guanidine (shown) is about as strong base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines.
(b) Show why p-nitroaniline is a much weaker base (3 pKb units weaker) than aniline.
(c) Explain why N,N-2,6-tetramethylaniline (shown) is a much stronger base than N,N-dimethylaniline.
Give the products expected when the following tertiary amines are treated with a peroxyacid and heated.
(c) cyclohexyl dimethyl amine (d) N-ethyl piperidine
Basicity depends on availability of an electron pair to bond a proton. Correlate structural effects in these amines with their basicities.
(a) Explain this order:
Question. (a) Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl amine (N-ethylpropan-1-amine), shown below.
(b) Show how a similar cleavage in the ethyl group gives an ion of m/z72.
(c) Explain why the peak at m/z 72 is much weaker than the one at m/z 58.
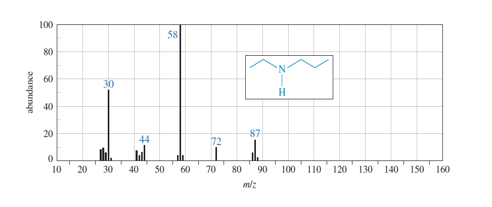
(a) Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl amine (N-ethylpropan-1-amine), shown below.
(b) Show how a similar cleavage in the ethyl group gives an ion of m/z72.
(c) Explain why the peak at m/z 72 is much weaker than the one at m/z 58.
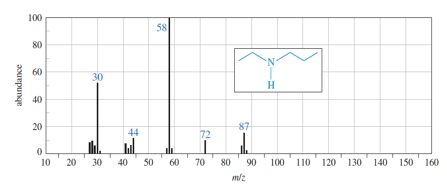
What do you think about this solution?
We value your feedback to improve our textbook solutions.