Chapter 19: Q24P (page 1014)
Propose a mechanism for the synthesis of methyl orange.
Short Answer
Step 1:
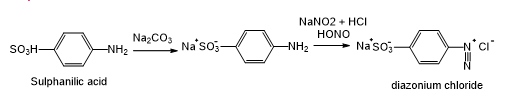
Step 2:
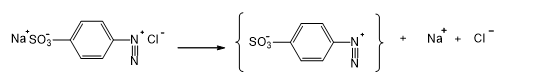
Step 3:
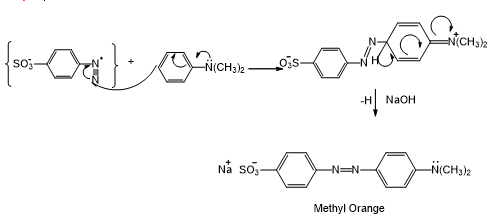
Learning Materials
Features
Discover
Chapter 19: Q24P (page 1014)
Propose a mechanism for the synthesis of methyl orange.
Step 1:

Step 2:

Step 3:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Propose a mechanism for the sulfonation of pyridine, and point out why sulfonation occurs at 3-position.
Rank each set of compounds in order of increasing basicity
(a) NaOH, NH3, CH3NH2, Ph-NH2
(b) aniline, p-methylaniline, p-nitroaniline
(c) aniline, pyrrole, pyridine, piperidine
(d) pyrrole, imidazole, 3-nitropyrrole
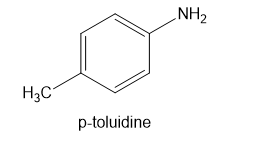
Show how p-toluidine can be converted to the following compounds, using any necessary reagents.
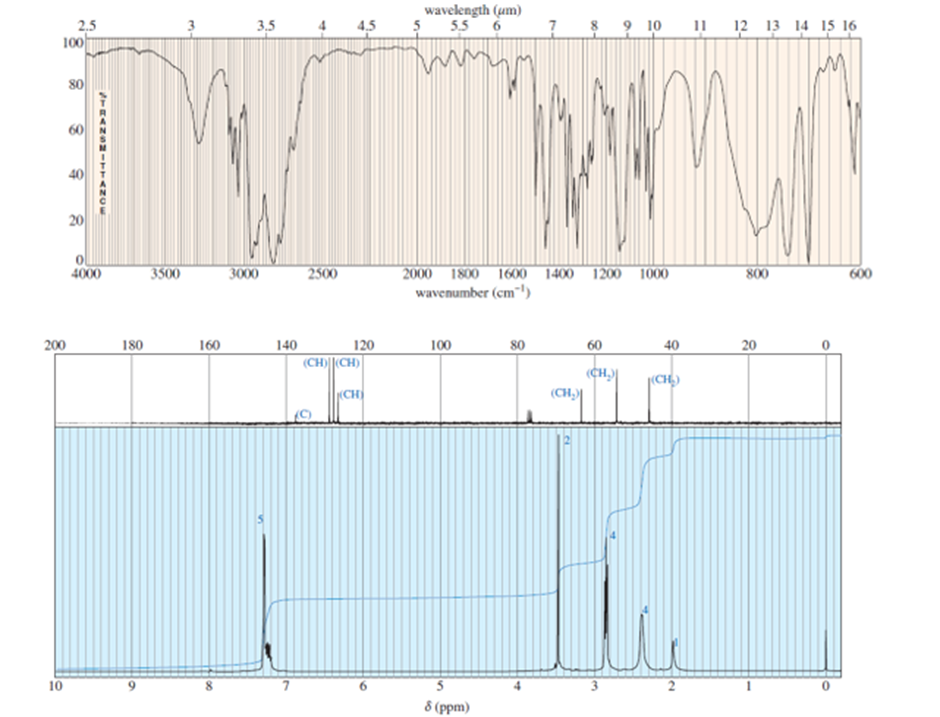
A compound of formula C11H16N2gives the IR, 1HNMR, and 13CNMR spectra shown. The proton NMR peak atδ 2.0disappears on shaking with D2O. Propose a structure for this compound, and show how your structure accounts for the observed absorptions.
Propose a mechanism for nitration of pyridine at the 4-position, and show why this orientation is not observed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.