Chapter 9: Q19P (page 484)
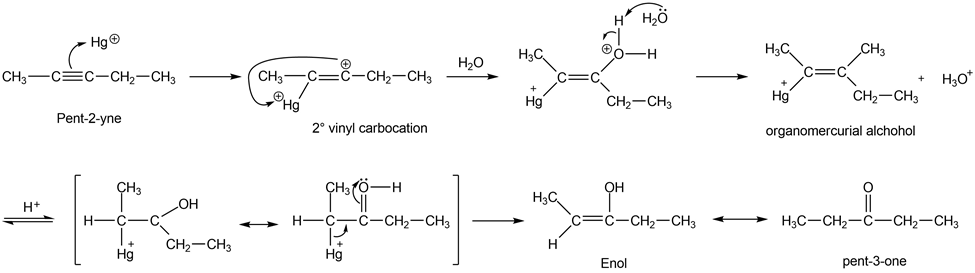
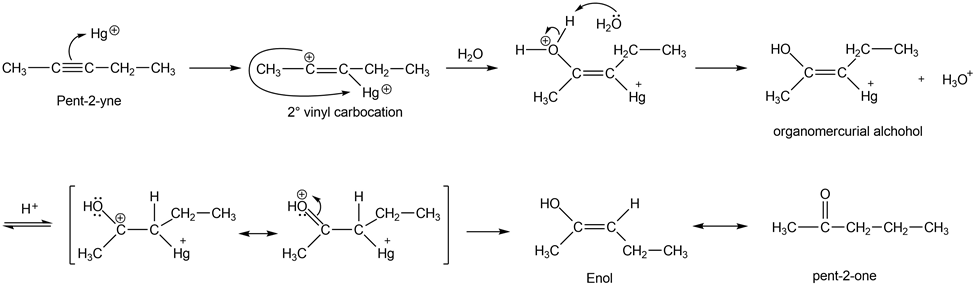
When pent-2-yne reacts with mercuric sulfate in dilute sulfuric acid, the product is a mixture of two ketones. Give the structures of these products, and use mechanisms to show how they are formed.
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q19P (page 484)
When pent-2-yne reacts with mercuric sulfate in dilute sulfuric acid, the product is a mixture of two ketones. Give the structures of these products, and use mechanisms to show how they are formed.


All the tools & learning materials you need for study success - in one app.
Get started for free
The fragrance of (Z)-1-phenylhex-2-en-1-ol resembles that of roses, with a delicate citrus edge. Show how you would synthesize this compound from benzaldehyde (PhCHO) and any other reagents you need.
Question:Show how you would accomplish the following synthetic transformations. Show all intermediates.
(��)2,2-�徱�����dz��Dz��ܳٲ��Ա�→b�ܳ�-1-��Ա�
(��)2,2-�徱�����dz��Dz��ܳٲ��Ա�→b�ܳ�-2-��Ա�
(��)���ܳ�-1-��Ա�→o����-3-��Ա�
(d)trans-����-2-��Ա�→h���-2-��Ա�
(��)2,2-�徱�����dz��dz��油�Ա�→h���-1-��Ա�
(��)���⳦���ǻ��Ա�→cis-cyclodecene
(��)���⳦���ǻ��Ա�→trans-cyclodecene
(h)hex-1-yne→hexan-2-one, CH3COCH2CH2CH2CH3
(i)hex-1-yne→hexanal, CH3(CH2)4CHO
(j)trans-����-2-��Ա�→cis-hex-2-ene
The boiling points of hex-1-ene (64 °C) and hex-1-yne (71 °C) are sufficiently close that it is difficult to achieve a clean separation by distillation. Show how you might use the acidity of hex-1-yne to remove the last trace of it from a sample of hex-1-ene.
Propose a mechanism for the reaction of pent-1-yne with HBr in the presence of peroxides. Show why anti-Markovnikov orientation results.
Question:When treated with hydrogen and a platinum catalyst, an unknown compound X absorbs 5 equivalents of hydrogen to give n-butylcyclohexane. Treatment of X with an excess of ozone, followed by dimethyl sulfide and water, gives the following products:
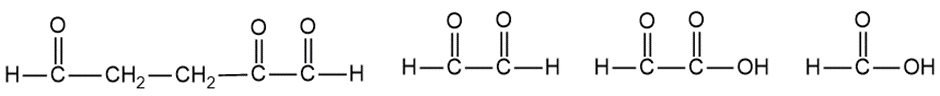
Propose a structure for the unknown compound X. Is there any uncertainty in your structure?
What do you think about this solution?
We value your feedback to improve our textbook solutions.