Chapter 9: Q18P (page 482)
Show how hex-1-yne might be converted to
(a) 1,2-dichlorohex-1-ene. (b) 1-bromohex-1-ene.
(c) 2-bromohex-1-ene. (d) 1,1,2,2-tetrabromohexane.
(e) 2-bromohexane. (f) 2,2-dibromohexane.
Short Answer
(a)
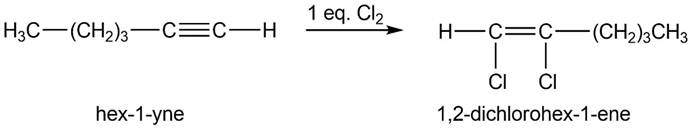
(b)
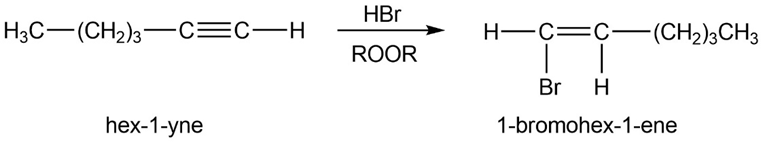
(c)
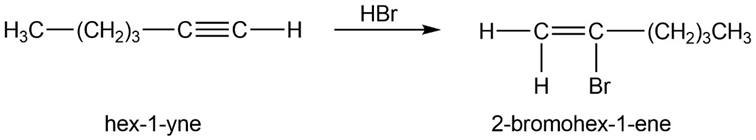
(d)
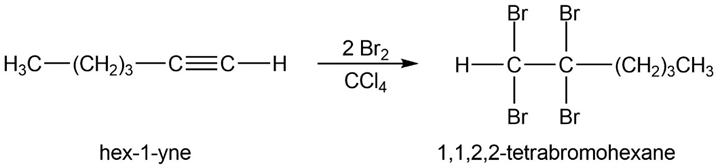
(e)

(f)

Learning Materials
Features
Discover
Chapter 9: Q18P (page 482)
Show how hex-1-yne might be converted to
(a) 1,2-dichlorohex-1-ene. (b) 1-bromohex-1-ene.
(c) 2-bromohex-1-ene. (d) 1,1,2,2-tetrabromohexane.
(e) 2-bromohexane. (f) 2,2-dibromohexane.
(a)

(b)

(c)

(d)

(e)

(f)

All the tools & learning materials you need for study success - in one app.
Get started for free
Deduce the structure of each compound from the information given. All unknown in this problem have molecular formula C8H12.
(a)Upon catalytic hydrogenation, unknown Wgives cyclooctane. Ozonolysis of W,followed by reduction with dimethylsulfide, gives octanedioic acid,HOOC-(CH2) 6-COOH . Draw the structure of W.
(b)Upon catalytic hydrogenation, unknown Xgives cyclooctane. Ozonolysis of X,followed by reduction with dimethyl sulfide, gives two equivalents of butanedial , O =CH -CH2 CH 2-CH = O. Draw the structure of X.
(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethyl sulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.
(d) Upon catalytic hydrogenation, unknown Z gives cis-bicyclo[4.2.0]octane. Ozonolysis of Z, followed by reduction with dimethyl sulfide, gives a cyclobutane with a three-carbon aldehyde (-CH2-CH2-CHO ) group on C1 and a one-carbon aldehyde (CHO) group on C2. Draw the structure of Z.
Question:The application box in the margin of page 473 states, “The addition of an acetylide ion to a carbonyl group is used in the synthesis of ethchlorvynol, a drug used to cause drowsiness and induce sleep.” Show how you would accomplish this synthesis from acetylene and a carbonyl compound.

ethchlorvynol
Question:Using hex-1-ene as your starting material, show how you would synthesize the following compounds. (Once you have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem)
(a)1,2-dibromohexane
(b)hex-1-yne
(c)2,2-dibromohexane
(d)hex-2-yne
(e)hexan-2-one
(f)hexanal
(g)pentanoic acid
(h)pentanal
(i)undec-6-yn-5-ol
The following functional group interchange is a useful synthesis of aldehydes.
(a)What reagents were used in this chapter for this transformation? Give an example to illustrate this method.
(b)This functional group interchange can also be accomplished using the following sequence.
Propose mechanisms from these steps.
(c) Explain why a nucleophilic reagent such as ethoxide adds to an alkyne more easily than it adds to an alkene.
Question:When treated with hydrogen and a platinum catalyst, an unknown compound X absorbs 5 equivalents of hydrogen to give n-butylcyclohexane. Treatment of X with an excess of ozone, followed by dimethyl sulfide and water, gives the following products:
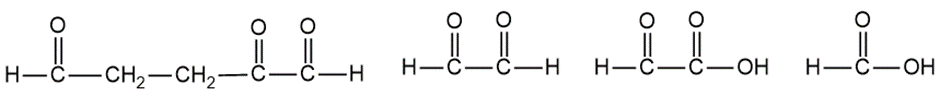
Propose a structure for the unknown compound X. Is there any uncertainty in your structure?
What do you think about this solution?
We value your feedback to improve our textbook solutions.