Chapter 9: Q17P (page 482)
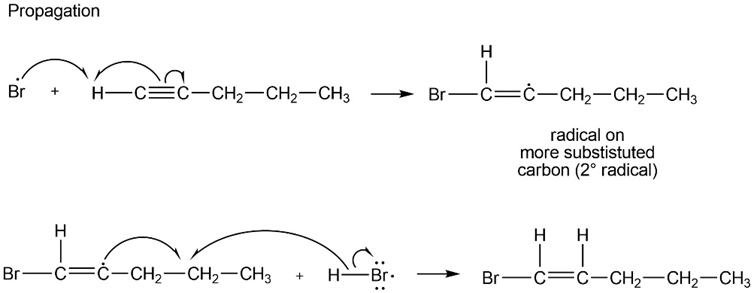
Propose a mechanism for the reaction of pent-1-yne with HBr in the presence of peroxides. Show why anti-Markovnikov orientation results.
Short Answer
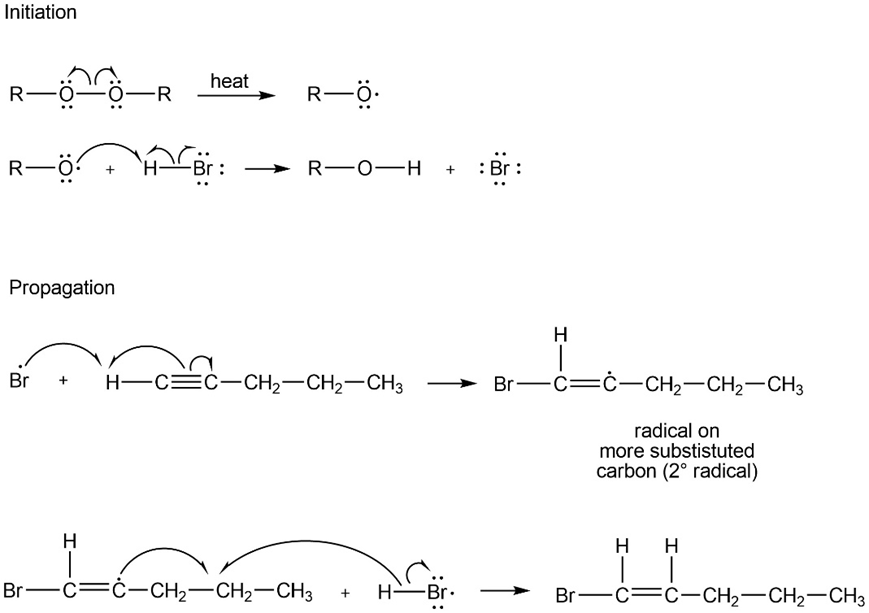
Anti-Markovnikov orientation results to form the more stablefree radical.
 Initiation step
Initiation step