Chapter 9: Q15P (page 482)
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov’s rule should be observed in both the first and second additions of HBr.
Learning Materials
Features
Discover
Chapter 9: Q15P (page 482)
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov’s rule should be observed in both the first and second additions of HBr.
All the tools & learning materials you need for study success - in one app.
Get started for free
The following functional group interchange is a useful synthesis of aldehydes.
(a)What reagents were used in this chapter for this transformation? Give an example to illustrate this method.
(b)This functional group interchange can also be accomplished using the following sequence.
Propose mechanisms from these steps.
(c) Explain why a nucleophilic reagent such as ethoxide adds to an alkyne more easily than it adds to an alkene.
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov’s rule should be observed in both the first and second additions of HBr.
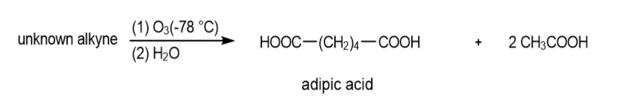
Oxidative cleavages can help to determine the positions of the triple bonds in alkynes.
(a) An unknown alkyne undergoes oxidative cleavage to give adipic acid and two equivalents of acetic acid. Propose a structure for the alkyne.
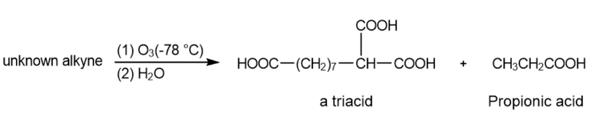
(b) An unknown alkyne undergoes oxidative cleavage to give the following triacid plus one equivalent of propionic acid. Propose a structure for the alkyne.
Question:When treated with hydrogen and a platinum catalyst, an unknown compound X absorbs 5 equivalents of hydrogen to give n-butylcyclohexane. Treatment of X with an excess of ozone, followed by dimethyl sulfide and water, gives the following products:

Propose a structure for the unknown compound X. Is there any uncertainty in your structure?
Question:The application box in the margin of page 473 states, “The addition of an acetylide ion to a carbonyl group is used in the synthesis of ethchlorvynol, a drug used to cause drowsiness and induce sleep.” Show how you would accomplish this synthesis from acetylene and a carbonyl compound.
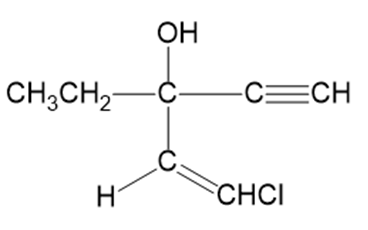
ethchlorvynol
What do you think about this solution?
We value your feedback to improve our textbook solutions.