Chapter 9: Q10P (page 476)
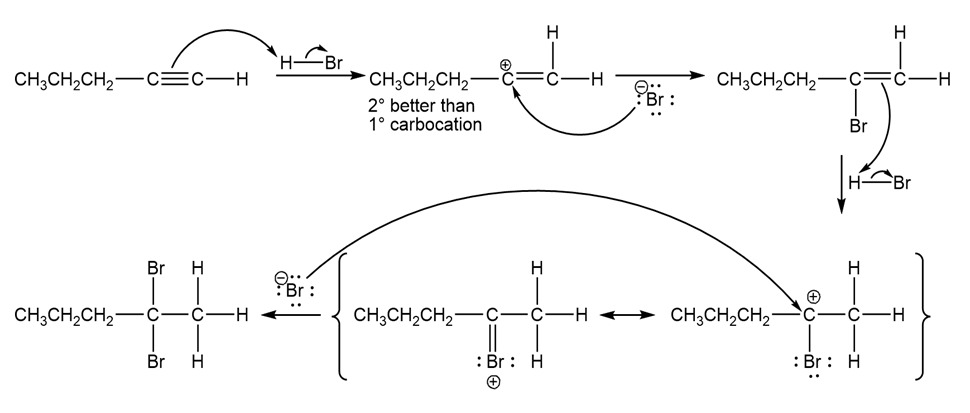
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov’s rule should be observed in both the first and second additions of HBr.
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q10P (page 476)
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov’s rule should be observed in both the first and second additions of HBr.

All the tools & learning materials you need for study success - in one app.
Get started for free
The hydroboration–oxidation of internal alkynes produces ketones.
(a) When hydroboration–oxidation is applied to but-2-yne, a single pure product is obtained. Determine the structure of this product, and show the intermediates in its formation.
(b) When hydroboration–oxidation is applied to pent-2-yne, two products are obtained. Show why a mixture of products should be expected with any unsymmetrical internal alkyne.
Predict the major product(s) of the following reactions:
(a) phenylacetylene + 2 HBr
(b) hex-1-yne + 2 HCl
(c) cyclooctyne + 2 HBr
(d) hex-2-yne + 2 HCl
Question: Show how you would synthesize each compound, beginning with acetylene and any necessary additional reagents.
(a) prop-2-yn-1-ol (propargyl alcohol)
(b) hept-2-yn-4-ol
(c) 2-phenylbut-3-yn-2-ol
(d) 3-methylhex-4-yn-3-ol
When pent-2-yne reacts with mercuric sulfate in dilute sulfuric acid, the product is a mixture of two ketones. Give the structures of these products, and use mechanisms to show how they are formed.
Question:When treated with hydrogen and a platinum catalyst, an unknown compound X absorbs 5 equivalents of hydrogen to give n-butylcyclohexane. Treatment of X with an excess of ozone, followed by dimethyl sulfide and water, gives the following products:
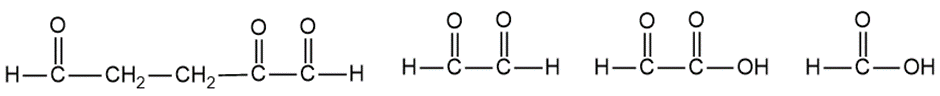
Propose a structure for the unknown compound X. Is there any uncertainty in your structure?
What do you think about this solution?
We value your feedback to improve our textbook solutions.