Chapter 6: 46P-d (page 330)
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
d)
Short Answer
d) The synthesis reaction of given compound is as follows,
Learning Materials
Features
Discover
Chapter 6: 46P-d (page 330)
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
d)
d) The synthesis reaction of given compound is as follows,
All the tools & learning materials you need for study success - in one app.
Get started for free
Triethyloxonium tetrafluoroborate, (CH3CH2)3O+ BF4−, is a solid with melting point 91–92°C. Show how this reagent can transfer an ethyl group to a nucleophile (Nuc:−) in an SN2 reaction. What is the leaving group? Why might this reagent be preferred to using an ethyl halide? (Consult Table 6-2.)
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
c)
Predict the major product of the following substitutions.
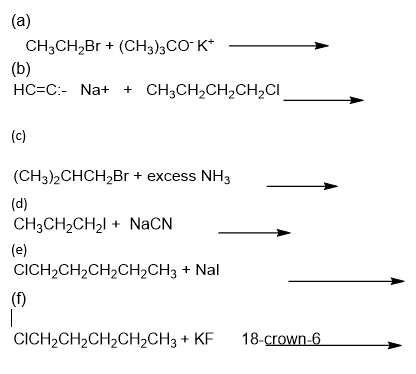
Give the structures of the following compounds.
(a) Methylene chloride
(b) Carbon tetrachloride
(c) 3-iodo-2-methylpentane
(d) Chloroform
(e) 2-chloro-3-ethyl-2-methyhexane
(f) Isobutyl iodide
(g) Cis-1-chloro-3-(chloromethyl)cyclohexane
(h) Tert-butyl bromide
For each of the following compounds,
What do you think about this solution?
We value your feedback to improve our textbook solutions.