Chapter 6: Q14P (page 300)
Predict the major product of the following substitutions.
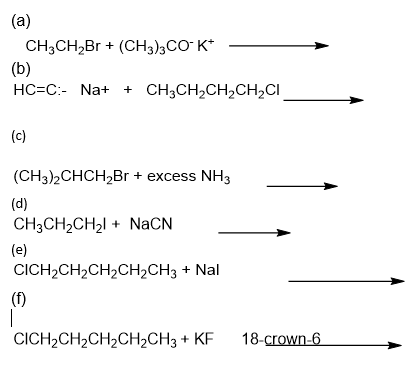
Short Answer
Answer
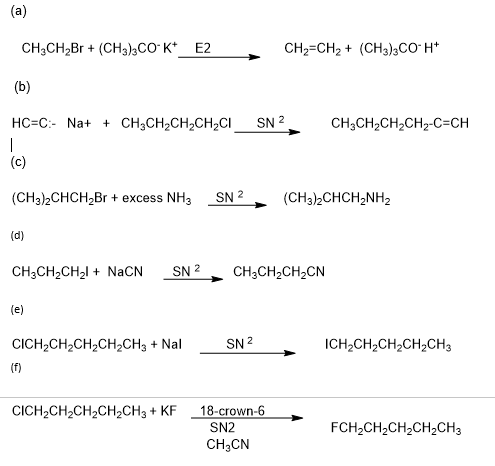
Learning Materials
Features
Discover
Chapter 6: Q14P (page 300)
Predict the major product of the following substitutions.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
e)
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning.
(a) ethyl chloride or ethyl iodide
(b) 1-bromopropane or cyclopropane
(c) cis-2,3-dibromobut-2-ene or trans-2,3-dibromobut-2-ene
(d) cis-1,2-dichlorocyclobutane or trans-1,3-dichlorocyclobutane.
Predict the compound in each pair that will undergo the SN2 reaction faster.
(a.)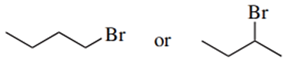
(b.)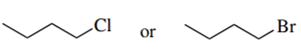
(c.)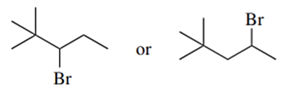
(d.)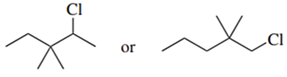
(e.)
(f.)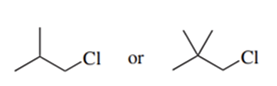
For each of the following compounds,
List the following carbocations in decreasing order of their stability.
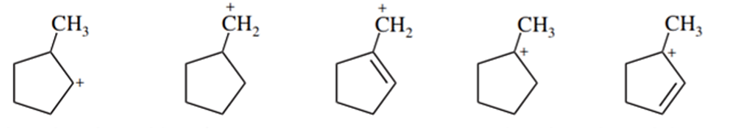
What do you think about this solution?
We value your feedback to improve our textbook solutions.