Chapter 6: 46P-c (page 330)
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
c)
Short Answer
c) The synthesis reaction of given compound is as follows,
Learning Materials
Features
Discover
Chapter 6: 46P-c (page 330)
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
c)
c) The synthesis reaction of given compound is as follows,
All the tools & learning materials you need for study success - in one app.
Get started for free
Give systematic (IUPAC) names for the following compounds.
Propose mechanisms to account for the observed products in the following reactions.
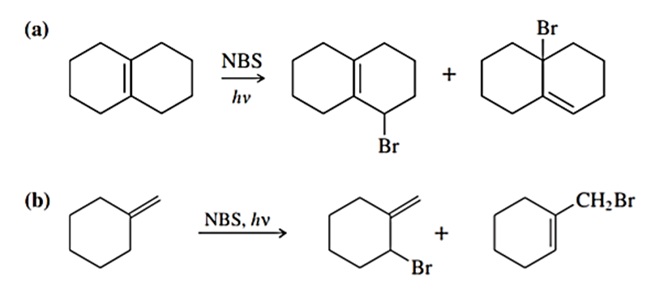
Classify each compound as an alkyl halide, a vinyl halide, or an aryl halide.
(a) (CH3)3CF
(b) CH3CHCBrCH3
(c) CH3CF3
(d)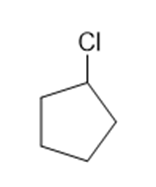
(e)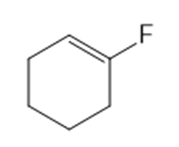
(f)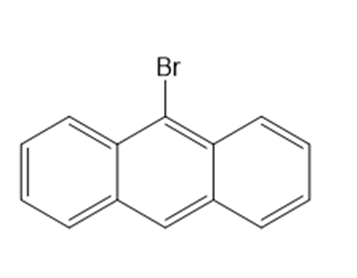
Question: Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds.
e)
For each pair of compounds, predict which one has the higher molecular dipole moment, and explain your reasoning.
(a) ethyl chloride or ethyl iodide
(b) 1-bromopropane or cyclopropane
(c) cis-2,3-dibromobut-2-ene or trans-2,3-dibromobut-2-ene
(d) cis-1,2-dichlorocyclobutane or trans-1,3-dichlorocyclobutane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.