Chapter 2: Q14P (page 117)
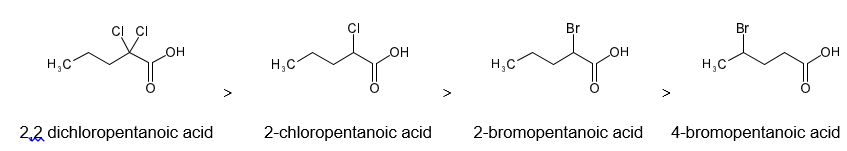
Rank the following acids in decreasing order of their acid strength. In each case, explain why the previous compound should be a stronger acid than the one that follows it.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q14P (page 117)
Rank the following acids in decreasing order of their acid strength. In each case, explain why the previous compound should be a stronger acid than the one that follows it.

All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethyl ether and ethanol are isomers. Their boiling points are very different, however. Explain why these two compounds have dramatically different boiling points.
CH3-O-CH3 CH3CH2-OH
dimethyl ether, bp -24 °C ethanol, bp 78 °C
The following compound can become protonated on any of the three nitrogen atoms. One of these nitrogens is much more basic than the others, however.
The following compounds can all react as bases.
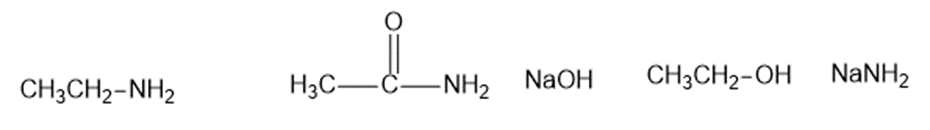
Predict the products of the following acid-base reactions.
(a)
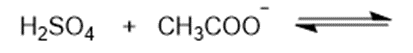
(b)
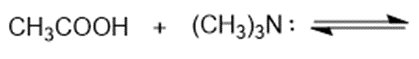
(c)
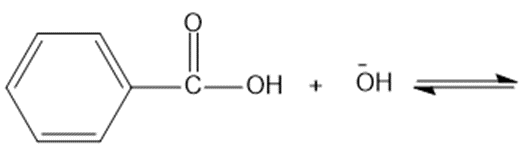
(d)
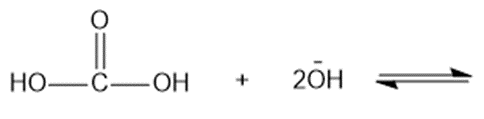
(e)

(f)
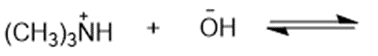
(g)
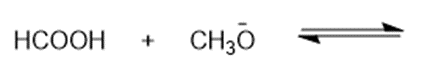
(h)
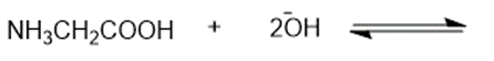
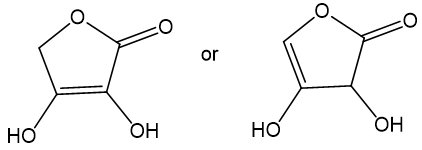
Choose the more acidic member of each pair of isomers, and show why the acid you chose is more acidic.
(a)
What do you think about this solution?
We value your feedback to improve our textbook solutions.