Chapter 27: Q 49. (page 1102)
Question: Draw structures for A, B, and C in the following reaction sequence and identify the process that converts B to C.
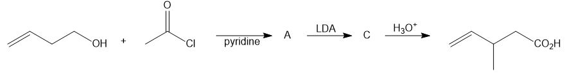
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 49. (page 1102)
Question: Draw structures for A, B, and C in the following reaction sequence and identify the process that converts B to C.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: For each molecular orbital in Figure 27.2, count the number of bonding interactions (interactions between adjacent orbitals of similar phase) and the number of nodes. (a) How do these two values compare for a bonding molecular orbital? (b) How do these two values compare for an antibonding molecular orbital?
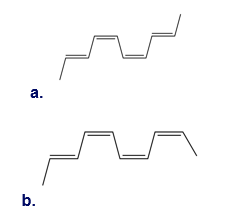
Question: What cyclic product is formed when each decatetraene undergoes thermal electrocyclic ring closure?
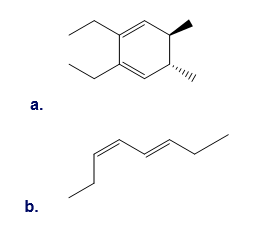
Question: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
Question:What starting materials are needed to synthesize each compound by a thermal [4 + 2] cycloaddition?
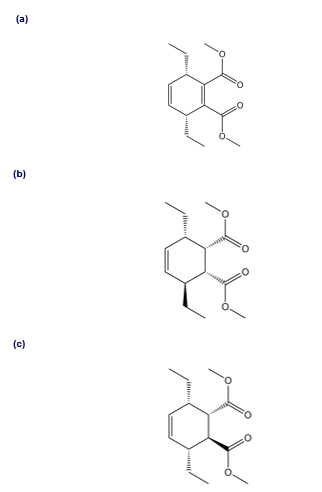
Question: Explain why heating buta-1,3-diene forms 4-vinylcyclohexene but not cycloocta-1,5-diene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.