Chapter 27: Q 32. (page 1100)
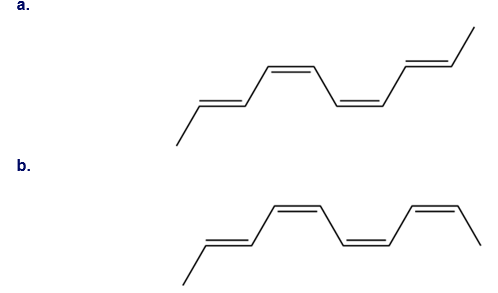
Question: What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring closure?
Short Answer
Answer
Formed cyclic product:
a. 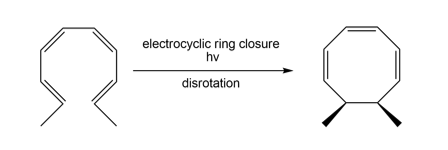
b. 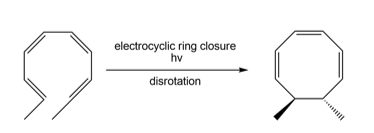
Learning Materials
Features
Discover
Chapter 27: Q 32. (page 1100)
Question: What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring closure?

Answer
Formed cyclic product:
a. 
b. 
All the tools & learning materials you need for study success - in one app.
Get started for free
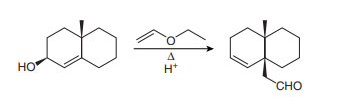
Question: Draw a stepwise, detailed mechanism for the following reaction.
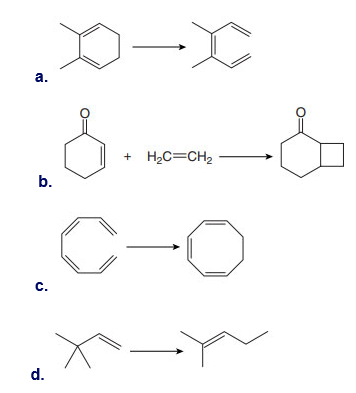
Question: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic rearrangement. Label the σ bonds that are broken or formed in each reaction.
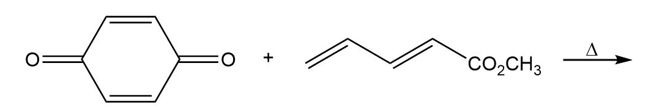
Question:(a) Draw the product of the following [4 + 2] cycloaddition, which was carried out in the early stages of the synthesis of the alkaloid reserpine (Problem 22.83). Indicate the stereochemistry at any newly formed stereogenic centers. (b) Draw the porbitals of the alkene and the terminal carbons of the conjugated diene, and show how the orientation of the reactants and orbital overlap lead to the observed stereochemistry.
Question: Using the Woodward–Hoffmann rules in Table 27.4, predict the stereochemistry of each reaction.
a. a [6 + 4] thermal cycloaddition
b. photochemical electrocyclic ring closure of deca-1,3,5,7,9-pentaene
c. a [4 + 4] photochemical cycloaddition
d. a thermal [5,5] sigmatropic rearrangement
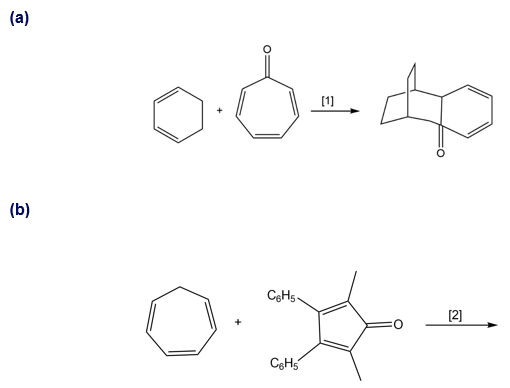
Question: What type of cycloaddition occurs in Reaction [1]? Draw the product of a similar process in Reaction [2]. Would you predict that these reactions occur under thermal or photochemical conditions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.