Chapter 27: Q 28. (page 1099)
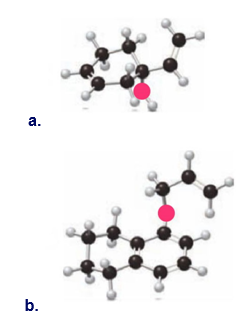
Question: What product is formed by the [3,3] sigmatropic rearrangement of each compound?
Short Answer
Answer
a. 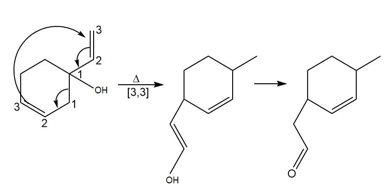
b.
Learning Materials
Features
Discover
Chapter 27: Q 28. (page 1099)
Question: What product is formed by the [3,3] sigmatropic rearrangement of each compound?

Answer
a. 

b.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Draw a stepwise mechanism for the Carroll rearrangement, a reaction that prepares aγ,δ-unsaturated carbonyl compound from aβ-keto ester and allylic alcohol in the presence ofbase.
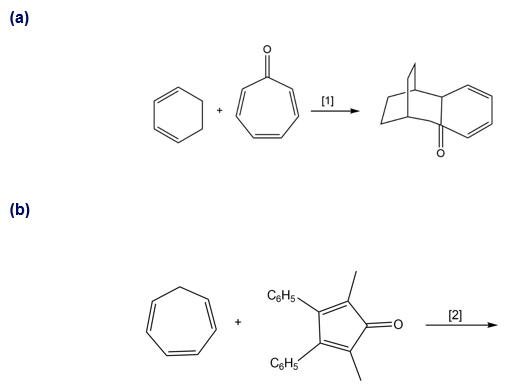
Question: What type of cycloaddition occurs in Reaction [1]? Draw the product of a similar process in Reaction [2]. Would you predict that these reactions occur under thermal or photochemical conditions?
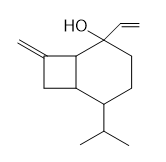
Question: One step in the synthesis of periplanone B, the chapter-opening molecule, involved anionic oxy-Cope rearrangement of the following unsaturated alcohol. Draw the product that results after protonation of the intermediate enolate.
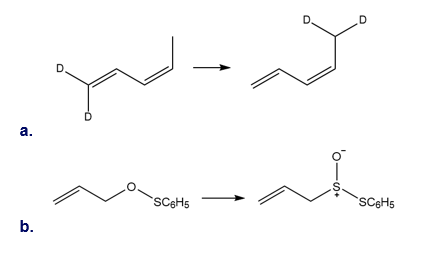
Question: What type of sigmatropic rearrangement is illustrated in each reaction?
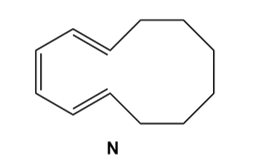
Question: a) What product is formed when triene N undergoes thermal electrocyclic ring closure? (b) What product is formed when triene N undergoes photochemical ring closure? (c) Label each process as conrotatory or disrotatory.
What do you think about this solution?
We value your feedback to improve our textbook solutions.