Chapter 13: Q 61. (page 526)
Question: Explain why a ketone carbonyl typically absorbs at a lower wavenumber than an aldehyde carbonyl (1715 vs. 1730cm-1)
Short Answer
Answer
Weaker bonds would vibrate at a lower frequency.
Learning Materials
Features
Discover
Chapter 13: Q 61. (page 526)
Question: Explain why a ketone carbonyl typically absorbs at a lower wavenumber than an aldehyde carbonyl (1715 vs. 1730cm-1)
Answer
Weaker bonds would vibrate at a lower frequency.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What is the mass of the molecular ion formed from compounds having each molecular formula:
(a) ; (b);(c); (d) methamphetamine ?
Question: The low-resolution mass spectrum of an unknown analgesic X had a molecular ion of 151. Possible molecular formulas include , , and . High-resolution mass spectrometry gave an exact mass of 151.0640. What is the molecular formula of X?
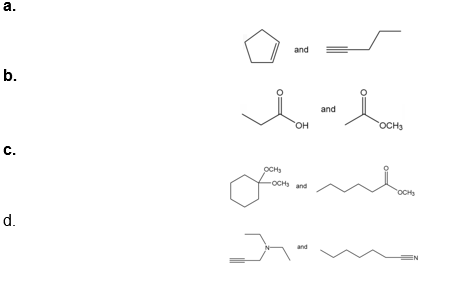
Question: How would each of the following pairs of compounds differ in their IR spectra?
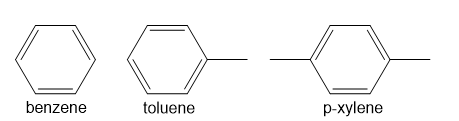
Question: Benzene, toluene, and p-xylene (BTX) are often added to gasoline to boost octane ratings. What would be observed if a mixture of these three compounds were subjected to GC–MS analysis? How many peaks would be present in the gas chromatogram? What would be the relative order of the peaks? What molecular ions would be observed in the mass spectra?
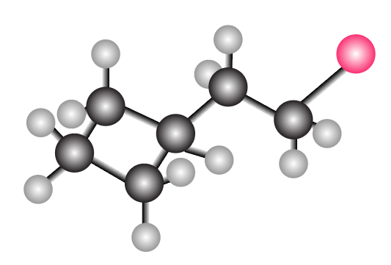
Question: What molecular ions would you expect for the compound depicted in the ball-and-stick model?
What do you think about this solution?
We value your feedback to improve our textbook solutions.