Chapter 31: Question 31.12 (page 1250)
Write a stepwise mechanism for the following reaction.
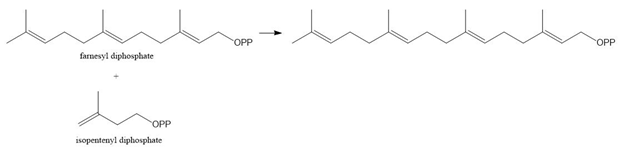
Short Answer
Answer
Learning Materials
Features
Discover
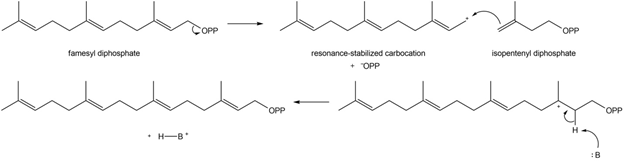
Chapter 31: Question 31.12 (page 1250)
Write a stepwise mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for the following reaction.
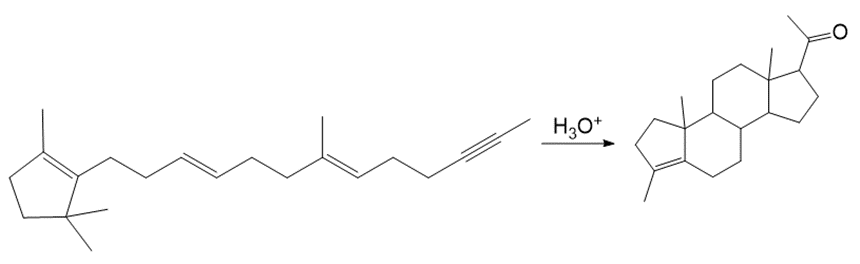
What is the structure of an optically inactive triacylglycerol that yields two moles of oleic acid and one mole of palmitic acid when hydrolyzed in aqueous acid?
One component of jojoba oil is a wax formed from eicosenoic acid and . Draw the structure of the wax, including the cis geometry of both carbon-carbon double bonds.
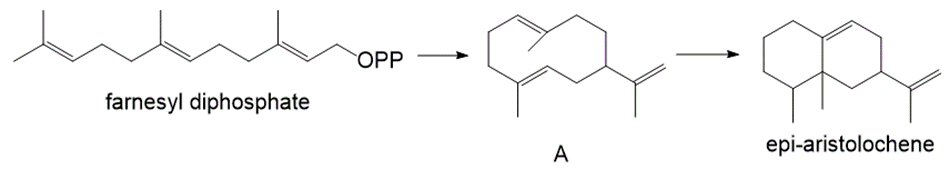
Farnesyl diphosphate is cyclized to sesquiterpene A, which is then converted to the bicyclic product epi-aristolochene. Write a stepwise mechanism for both reactions.
Phosphoacylglycerols should remind you of soaps (Section 3.6). In what ways are these compounds similar?
What do you think about this solution?
We value your feedback to improve our textbook solutions.