Chapter 19: Q42E (page 1096)
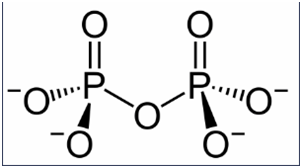
Explain how the diphosphate ion, \({\left( {{O_3}P - O - P{O_3}} \right)^{4 - }}\) can function as a water softener that prevents the precipitation of \(F{e^{2 + }}\) as an insoluble iron salt.
Short Answer
Due to its structure, oxygen on one side can form bonds with water and on the other one with \(F{e^{2 + }}\) as it is bonding with water, \(F{e^{2 + }}\) will remain insoluble.