Chapter 20: Q56E (page 1145)
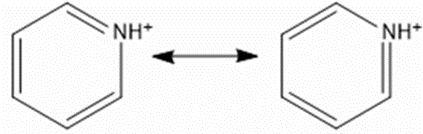
Write the two resonance structures for the pyridinium ion, \({C_5}{H_5}N{H^ + }. \)
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q56E (page 1145)
Write the two resonance structures for the pyridinium ion, \({C_5}{H_5}N{H^ + }. \)

All the tools & learning materials you need for study success - in one app.
Get started for free
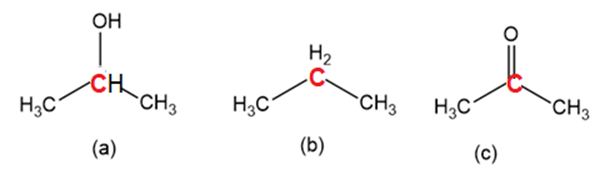
Order the following molecules from least to most oxidized, based on the marked carbon atom:
Write two complete balanced equations for the following reaction, one using condensed formulas and one using Lewis structures.
Methyl amine is added to a solution of \(HCl.\)
Write Lewis structures and name the five structural isomers of hexane.
Give the complete IUPAC name for each of the following compounds:
What is the molecular structure about the nitrogen atom in trimethyl amine and in the trimethyl ammonium ion, \({\left( {C{H_3}} \right)_3}N{H^ + }\)? What is the hybridization of the nitrogen atom in trimethyl amine and in the trimethyl ammonium ion?
What do you think about this solution?
We value your feedback to improve our textbook solutions.