Chapter 20: Q54E (page 1145)
Write the Lewis structures of both isomers with the \(formula {C_2}{H_7}\;N. \)
Short Answer
Two isomers are shown in the pictures.
On the left, there is dimethyl amine. On the right, it's ethyl amine.
Learning Materials
Features
Discover
Chapter 20: Q54E (page 1145)
Write the Lewis structures of both isomers with the \(formula {C_2}{H_7}\;N. \)
Two isomers are shown in the pictures.
On the left, there is dimethyl amine. On the right, it's ethyl amine.
All the tools & learning materials you need for study success - in one app.
Get started for free
MTBE, Methyl tert-butyl ether, CH3OC(CH3)3, is used as an oxygen source in oxygenated gasolines. MTBE is manufactured by reacting 2-methylpropene with methanol.
(a) Using Lewis structures, write the chemical equation representing the reaction.
(b) What volume of methanol, density 0.7915 g/mL, is required to produce exactly 1000 kg of MTBE, assuming a 100% yield?
Explain why it is not possible to prepare a ketone that contains only two carbon atoms.
Give the complete IUPAC name and the common name for each of the following compounds:
a)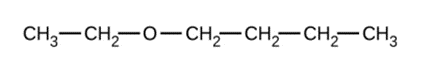
b)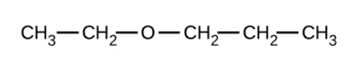
c)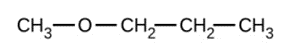
Write the two-resonance structures for the acetate ion.
Ethylene can be produced by the pyrolysis of ethane:
\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{6}}}\to{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{4}}}{\rm{+}}{{\rm{H}}_{\rm{2}}}\]
How many kilograms of ethylene is produced by the pyrolysis of 1.000 ×103 kg of ethane, assuming a 100.0%yield?
What do you think about this solution?
We value your feedback to improve our textbook solutions.