Chapter 20: Q27 E (page 1141)
What mass of 2-bromopropane could be prepared from 25.5 g of propene? Assume a 100% yield of product.
Short Answer
The mass of 2-bromopropane formed is 74.5 g.
Learning Materials
Features
Discover
Chapter 20: Q27 E (page 1141)
What mass of 2-bromopropane could be prepared from 25.5 g of propene? Assume a 100% yield of product.
The mass of 2-bromopropane formed is 74.5 g.
All the tools & learning materials you need for study success - in one app.
Get started for free
How does the hybridization of the substituted carbon atom change when alcohol is converted into an aldehyde? An aldehyde to a carboxylic acid?
Identify any carbon atoms that change hybridization and the change in hybridization during the reactions in Exercise 20.51.
Write structures for the three isomers of the aromatic hydrocarbon xylene,\({C_6}{H_4}{\left( {C{H_3}} \right)_2}\)
Write two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures. (a) 2-butene is treated with water in dilute acid (b) ethanol is dehydrated to yield ethene
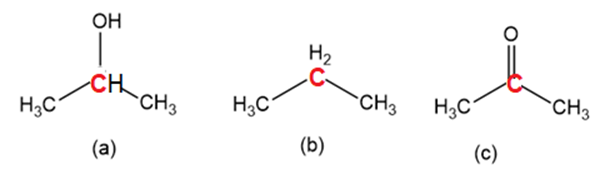
Order the following molecules from least to most oxidized, based on the marked carbon atom:
What do you think about this solution?
We value your feedback to improve our textbook solutions.