Chapter 20: Q20.2 CYL (page 1101)
Identify the chemical formula of the molecule represented here:
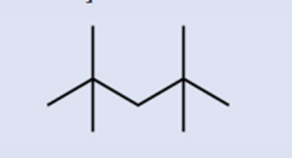
Short Answer
The given molecule has \({C_9}{H_{20}}\)as its chemical formula.
Learning Materials
Features
Discover
Chapter 20: Q20.2 CYL (page 1101)
Identify the chemical formula of the molecule represented here:

The given molecule has \({C_9}{H_{20}}\)as its chemical formula.
All the tools & learning materials you need for study success - in one app.
Get started for free
Benzene is one of the compounds used as an octane enhancer in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2H2 C6H6
Draw Lewis structures for these compounds, with resonance structures as appropriate, and determine the hybridization of the carbon atoms in each.
Draw Lewis structures for pyridine and its conjugate acid, the pyridinium ion, \({C_5}{H_5}N{H^ + }.\) \(What are\) the geometries and hybridizations about the nitrogen atoms in pyridine and in the pyridinium ion?
Write two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
(a) 1-butanol reacts with acetic acid
(b) propionic acid is poured onto solid calcium carbonate
Give the complete IUPAC name for each of the following compounds:
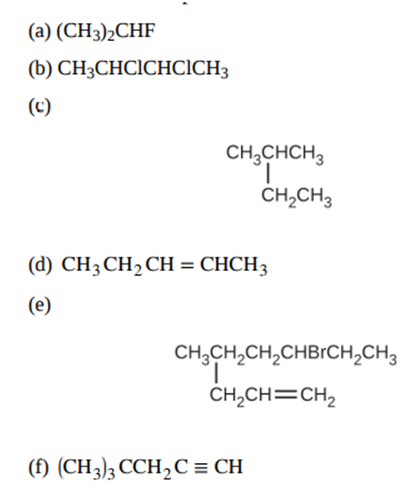
Write the condensed structures of all isomers with the formulaC2H6O2. Label the functional group (or groups) of each isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.