Chapter 10: Q96E (page 593)
Rubidium iodide crystallizes with a cubic unit cell that contains iodide ions at the corners and a rubidium ion in the center. What is the formula of the compound?
Short Answer
Molecular formula of rubidium iodide is Rbl.
Learning Materials
Features
Discover
Chapter 10: Q96E (page 593)
Rubidium iodide crystallizes with a cubic unit cell that contains iodide ions at the corners and a rubidium ion in the center. What is the formula of the compound?
Molecular formula of rubidium iodide is Rbl.
All the tools & learning materials you need for study success - in one app.
Get started for free
The boiling point of ethyl ether was measured to be 10 °C at a base camp on the slopes of Mount Everest. as shown to determine the approximate atmospheric pressure at the camp.
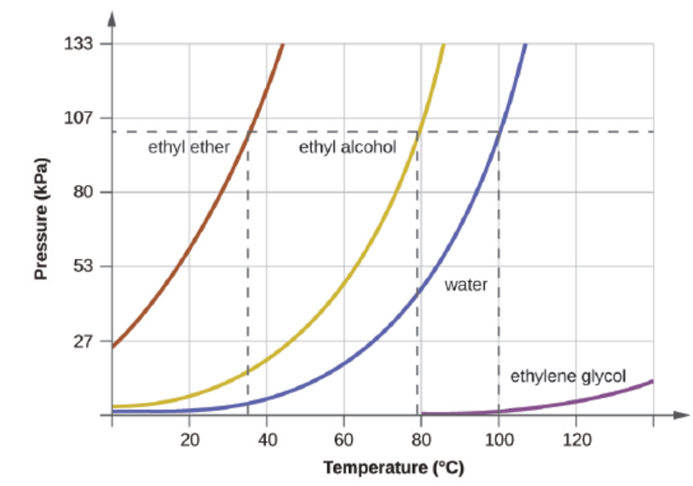
Figure 10.24 The boiling points of liquids are the temperatures at which their equilibrium vapor pressures equal the pressure of the surrounding atmosphere. Normal boiling points are those corresponding to a pressure of 1 atm (101.3
kPa.)
The white pigment titanium oxide crystallizes with titanium ions in one-half of the octahedral holes in a closest-packed array of oxide ions. What is the formula of titanium oxide?
The types of intermolecular forces in a substance are identical whether it is a solid, a liquid, or a gas. Why then does a substance change phase from a gas to a liquid or to a solid?
At 250C, how high will water rise in a glass capillary tube with an inner diameter of 0.63 mm?
The enthalpy of vaporization of \({\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{(l)}}\) is 9.8 kJ/mol. Would you expect the enthalpy of vaporization of \({\rm{C}}{{\rm{S}}_{\rm{2}}}{\rm{(l)}}\) to be 28 kJ/mol, 9.8 kJ/mol, or -8.4 kJ/mol? Discuss the plausibility of each of these answers.
What do you think about this solution?
We value your feedback to improve our textbook solutions.