Chapter 10: Q80E. (page 592)
Nickel metal crystallizes in a cubic closest packed structure. What is the coordination number of a nickel atom?
Short Answer
The coordination number of nickel in the cubic closest packing structure is 12.
Learning Materials
Features
Discover
Chapter 10: Q80E. (page 592)
Nickel metal crystallizes in a cubic closest packed structure. What is the coordination number of a nickel atom?
The coordination number of nickel in the cubic closest packing structure is 12.
All the tools & learning materials you need for study success - in one app.
Get started for free
A syringe at a temperature of \({\rm{2}}{{\rm{0}}^{\rm{o}}}{\rm{C}}\) is filled with liquid ether in such a way that there is no space for any vapour. If the temperature is kept constant and the plunger is withdrawn to create a volume that can be occupied by vapour, what would be the approximate pressure of the vapour produced?
Explain why a hydrogen bond between two water molecules is weaker than a hydrogen bond between two hydrogen fluoride molecules.
The boiling point of ethyl ether was measured to be 10 °C at a base camp on the slopes of Mount Everest. as shown to determine the approximate atmospheric pressure at the camp.
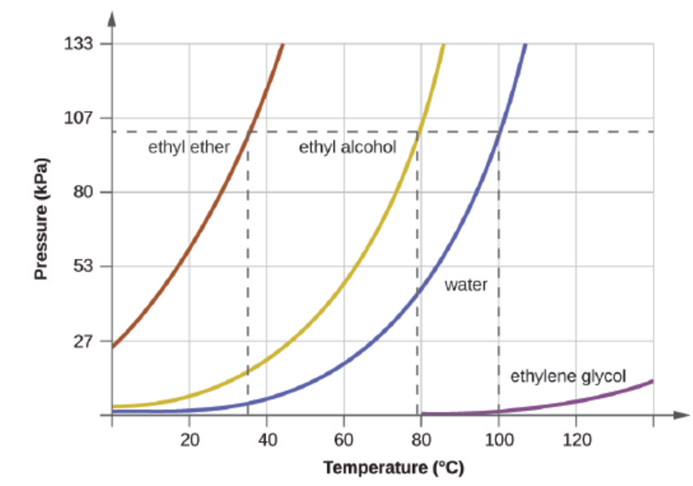
Figure 10.24 The boiling points of liquids are the temperatures at which their equilibrium vapor pressures equal the pressure of the surrounding atmosphere. Normal boiling points are those corresponding to a pressure of 1 atm (101.3
kPa.)
Ethyl chloride is used as a local anesthetic. When the liquid is sprayed on the skin, it cools the skin enough to freeze and numb it. Explain the cooling effect of liquid ethyl chloride.
The hydrogen fluoride molecule, HF, is more polar than a water molecule, \({{\rm{H}}_{\rm{2}}}{\rm{O}}\) (for example, HF has a greater dipole moment). Yet the molar enthalpy of vaporization for liquid hydrogen fluoride is lesser than that of water. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.