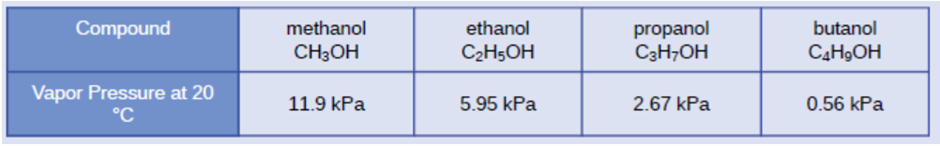
Chapter 10: Q13E (page 584)
On the basis of intermolecular attractions, explain the differences in the boiling points of n-butane \(\left( { - 1 ^\circ C} \right)\) and chloroethane \(\left( {12 ^\circ C} \right),\)which have similar molar masses.
Short Answer
Chloroethane has a higher boiling point than \({\rm{n}} - butane\) although they have similar masses.