The reaction
- The temperature is \({20^\circ }{\rm{C}}\)
- The pressure of \({\rm{C}}{{\rm{O}}_{\rm{2}}}\)in a closed cylinder at equilibrium is \({\rm{65\;atm}}\)
Let us sketch a plot with the change in pressure over time, as gaseous \({\rm{C}}{{\rm{O}}_{\rm{2}}}\) is released (the temperature is constant).
In the beginning, the vapour and liquid phases of carbon dioxide are in balance. The pressure is kept at \({\rm{65}}\) atmospheres. The system's pressure drops after the gaseous carbon dioxide is discharged.
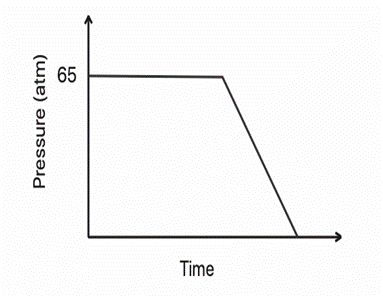
Hence, when some gaseous \({\rm{C}}{{\rm{O}}_{\rm{2}}}\)is released, the equilibrium of the system will move to the right, and so some liquid \({\rm{C}}{{\rm{O}}_{\rm{2}}}\)will convert into gas, to maintain the equilibrium pressure of \({\rm{65\;atm}}\).So, as long as we have liquid \({\rm{C}}{{\rm{O}}_{\rm{2}}}\),the pressure will be \({\rm{65\;atm}}\).As soon as we convert all liquid \({\rm{C}}{{\rm{O}}_{\rm{2}}}\)into gas, the pressure will begin to drop.




