Chapter 9: Q8 E (page 506)
During the Viking landings on Mars, the atmospheric pressure was determined to be on average about 6.50 millibars (1 bar = 0.987 atm). What is that pressure in torr and kPa?
Short Answer

Learning Materials
Features
Discover
Chapter 9: Q8 E (page 506)
During the Viking landings on Mars, the atmospheric pressure was determined to be on average about 6.50 millibars (1 bar = 0.987 atm). What is that pressure in torr and kPa?

All the tools & learning materials you need for study success - in one app.
Get started for free
One way to state Boyle’s law is “All other things being equal, the pressure of a gas is inversely proportional to its volume.” (a) What is the meaning of the term “inversely proportional?” (b) What are the “other things” that must be equal?
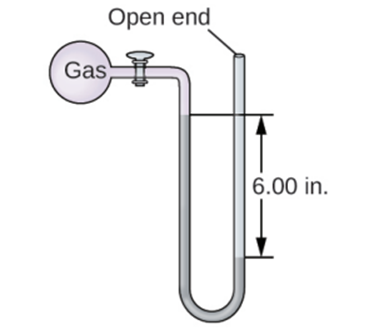
The pressure of a sample of gas is measured with an open-end manometer, partially shown to the right. The liquid in the manometer is mercury. Assuming atmospheric pressure is 29.92 in. Hg, determine the pressure of the gas in: (a) torr (b) Pa (c) bar.
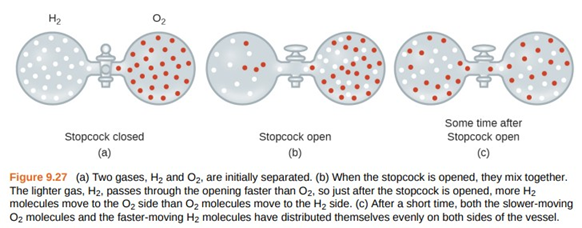
Question: Explain why the numbers of molecules are not identical in the left- and right-hand bulbs shown in the centre illustration of Figure \({\rm{9}}{\rm{.27}}\).
A commercial mercury vapor analyzer can detect, in air, concentrations of gaseous Hg atoms (which are poisonous) as low as 2×10-6mg/L of air. At this concentration, what is the partial pressure of gaseous mercury if the atmospheric pressure is 733 torr at 26 ̊C?
Most mixtures of hydrogen gas with oxygen gas are explosive. However, a mixture that contains less than 3.0 % O2 is not. If enough O2 is added to a cylinder of H2 at 33.2 atm to bring the total pressure to 34.5 atm, is the mixture explosive?
What do you think about this solution?
We value your feedback to improve our textbook solutions.