Chapter 9: Q14 E (page 506)
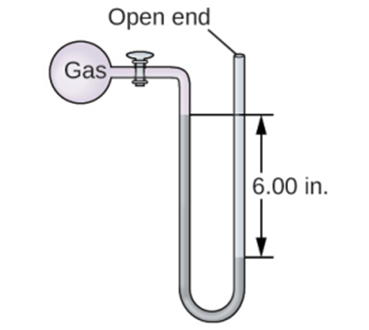
The pressure of a sample of gas is measured with an open-end manometer, partially shown to the right. The liquid in the manometer is mercury. Assuming atmospheric pressure is 29.92 in. Hg, determine the pressure of the gas in: (a) torr (b) Pa (c) bar.
Short Answer
- Pressure = 864.77 Torr
- Pressure = 112,893.06 Pa
- Pressure = 0.11287 bar