Chapter 9: Q50E (page 509)
Which is denser at the same temperature and pressure, dry air or air saturated with water vapour? Explain.
Short Answer
Because wet air has a lower molar mass than dry air, we can argue that wet air has a lower density.
Learning Materials
Features
Discover
Chapter 9: Q50E (page 509)
Which is denser at the same temperature and pressure, dry air or air saturated with water vapour? Explain.
Because wet air has a lower molar mass than dry air, we can argue that wet air has a lower density.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A balloon filled with helium gas is found to take6hours to deflate to50%of its original volume. How long will it take for an identical balloon filled with the same volume of hydrogen gas (instead of helium) to decrease its volume by 50%?
Sometimes leaving a bicycle in the sun on a hot day will cause a blowout. Why?
How would the graph in Figure 9.12 change if the number of moles of gas in the sample used to determine the curve were doubled?
One way to state Boyle’s law is “All other things being equal, the pressure of a gas is inversely proportional to its volume.” (a) What is the meaning of the term “inversely proportional?” (b) What are the “other things” that must be equal?
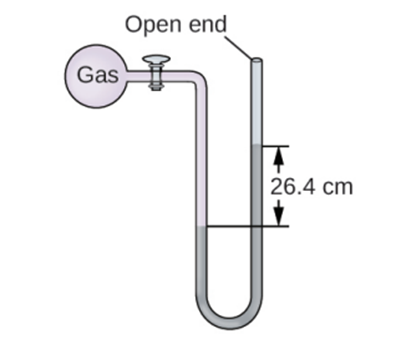
The pressure of a sample of gas is measured at sea level with an open-end mercury manometer. Assuming atmospheric pressure is 760.0mm Hg ,determine the pressure of the gas in: (a) mm Hg (b) (c) kPa.
What do you think about this solution?
We value your feedback to improve our textbook solutions.