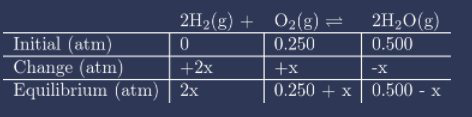Chapter 13: Q93E (page 761)
Question: Consider the reaction between \({{\rm{H}}_2}\)and \({{\rm{O}}_2}\)at 100 K\({K_P} = \frac{{{{\left( {{P_{{{\rm{H}}_2}{\rm{O}}}}} \right)}^2}}}{{\left( {{P_{{{\rm{O}}_2}}}} \right){{\left( {{P_{{{\rm{H}}_2}}}} \right)}^2}}} = 1.33 \times {10^{20}}\)
If 0.500 atm of H2 and 0.500 atm of O2are allowed to come to equilibrium at this temperature, what are the partial pressures of the components?
Short Answer
The partial pressure of \({{\rm{H}}_2}\)\({\rm{\;is\;}}8.67 \times {10^{ - 11}}{\rm{M}}\)
The partial pressure of \({{\rm{O}}_2}\)\({\rm{\;is\;}}0.250{\rm{M}}\)
The partial pressure of \({{\rm{H}}_2}{\rm{O\;is\;}}0.500{\rm{M}}\)